Interpretation
Septic suppurative inflammation
Explanation
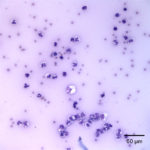
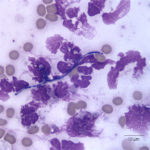
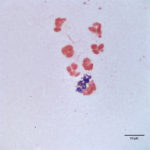
The direct smears of the joint fluid contained many cells consisting of 97% degenerate and dying neutrophils with 3% macrophages in a proteinaceous background. There was no evidence of windrowing (lining up of cells in rows), supporting decreased viscosity. Numerous gram positive cocci in pairs and chains were found extra- and intracellularly within neutrophils (Figure 2a, 3). A few aggregates of neutrophils were also seen (these aggregates may have mildly decreased the provided nucleated cell count). The morphologic features of the bacteria were compatible with Streptococcus species. The fluid was classified as an exudate (inflammatory synovitis, Question 1), secondary to bacterial infection.
 |
 |
 |
Additional Information
Anaerobic culture did not yield any growth but Streptococcus zooepidemicus was isolated from aerobic culture. The bacterium was susceptible to most of the tested antibiotics on sensitivity testing. The foal was treated with penicillin and amikacin along with gastroprotectants, probiotics and two serial plasma transfusions until the serum IgG concentration was higher than 800 mg/dL. Salmonella culture and Rotavirus antigen detection were negative.
Discussion
An inflammatory joint is defined as an increased proportion of neutrophils in the synovial fluid (usually >10% of total cells, particularly when associated with an increased total nucleated cell count, i.e. >500-1000 cells/uL)1. An inflammatory process in a joint leads us to three main disease processes: infection (septic joint), acute trauma, or immune-mediated disease. Trying to differentiate between these categories is challenging, especially when a pathogen is not identified on cytologic assessment or culture, which is the case with many septic joints in animals. Unlike small animals, particularly dogs, immune-mediated joint disease is rarely diagnosed in horses1. This may reflect a true low incidence of disease or our inability to confirm an immune-mediated process in this species due to the lack of diagnostic tests that are typically available for dogs (e.g. antinuclear antibody and rheumatoid factor testing). Therefore, our top differential diagnosis when a joint has an inflammatory component (with or without degenerate neutrophils) in horses should be bacterial sepsis. The second differential diagnosis would be acute non-septic trauma, which can also increase neutrophil counts, although not usually to the degree seen in this foal (Question 2).
The routes of joint infection are hematogenous spread of bacteria with systemic sepsis (most common mechanism in foals), direct extension from an adjacent soft tissue or bone infection, inoculation of the joint secondary to trauma, or iatrogenic2. A polyarthritis is more likely due to hematogenous spread, whereas monoarthritis usually indicates a localized problem, such as trauma or direct extension. The most common clinical signs associated with a septic joint in foals and horses are acute moderate to severe non-weight bearing lameness, joint swelling and pain2,3. In foals with septic arthritis, the tarsal joint is the most commonly affected site, with the hip joint being a rare site of infection2,4. In this foal, the suspected route of infection of the hip joint was the recent trauma (Question 2). Septic arthritis requires extended antibiotic treatment, can be associated with a poor prognosis and can decrease athlete potential in young performance horses2.
The most common reported blood abnormalities in foals with septic arthritis are a leukocytosis due to a moderate to marked neutrophilia2 and increased acute phase proteins such as SAA and fibrinogen5. These changes were evident in the foal of this report. One retrospective study evaluated the sensitivity and specificity of high fibrinogen concentrations as an indicator of physeal or epiphyseal osteomyelitis in foals with septic arthritis5. Of foals in which bone involvement was confirmed by radiographic findings, 94% (16/17) had very high fibrinogen concentrations (>900 mg/dL), with concentrations being lower in foals with septic arthritis without physeal or epiphyseal involvement (< 800 mg/dL). However, this study was relatively small and high fibrinogen concentrations just indicate systemic inflammation (3 foals with septic arthritis and no radiographic evidence of bone involvement had fibrinogen concentrations > 900 mg/dL). Another retrospective study of 83 foals with septic joints found that foals with higher fibrinogen concentrations (median 600 mg/dL) were more likely to survive to discharge than those with lower fibrinogen concentrations (median 465 mg/dL). However, there was substantial overlap between groups and the medical records were not assessed for radiographic changes of osteomyelitis4. Both studies raise questions about the diagnostic utility of fibrinogen measurement, particularly with a crude assay such as heat precipitation, and fibrinogen concentrations should not be relied upon for confirming bone involvement. Documenting concurrent osteomyelitis in foals with septic arthritis may be important because foals with physeal or epiphyseal involvement were more likely to be euthanized in the former study (2.6x higher risk, although confidence intervals were quite large, ranging from 0.53 to 13x)5. Similarly, another retrospective study of 81 foals showed that osteomyelitis was associated with decreased survival2. Involvement of more than 1 joint has also been related to poor outcome2,4, however it is possible that this may be related to the owners being given a poor prognosis, particularly for future performance of racing animals. The anemia in the foal of this report was attributed to inflammatory disease because there was evidence of systemic inflammation (high SAA, fibrinogen and peripheral neutrophil count). The microcytosis was an age-expected finding (normal for young foals due to a physiologic iron deficiency).
A septic joint can be diagnosed based on the combination of history and clinical signs (see above) and cytologic examination of the joint fluid. Microscopic evaluation of a septic joint fluid usually shows inflammatory changes such as an increased total nucleated cell count (usually >5000 cells/uL), comprised primarily of neutrophils, increased total protein (usually >2.5 g/dL) and decreased viscosity2,6. The decrease in joint viscosity is believed to be due to bacterial production of hyaluronidase, which degrades hyaluronic acid6. Neutrophils may or may not be degenerate and are typically non-degenerate in adult horses and degenerate in foals (as they were in this case), for unknown reasons. In one retrospective study, foals with degenerate neutrophils in the joint fluid were more likely to not survive2. On cytologic examination, it is common to not be able to find a pathogen, particularly in adult horses, therefore, the absence of a pathogen (or degenerate neutrophils) on cytologic examination does not rule out an infection. Bacteria may or may not be cultured from fluids with septic arthritis, but this should always be performed in suspected cases (Question 3). A culture will yield a definitive diagnosis about a possible pathogen, however bacterial culture of synovial fluid has a low sensitivity, ranging from 45% to 78% in several studies2,4,7,8. Culture of the synovial membrane has not proven to be more sensitive than synovial fluid culture alone9, arguing against this practice. In this case, bacteria were identified in the fluid by cytologic examination and were cultured from the joint fluid of the foal. Synovial glucose concentrations are not frequently measured but results can be informative. Synovial glucose concentrations should normally be similar to serum, since synovial fluid is mainly an ultra-filtrate for serum that is modified by the addition of hyaluronic acid from synoviocytes. Glycolytic activity of neutrophils and bacteria may result in a decreased synovial fluid glucose concentration compared to blood. A difference in the synovial to blood glucose concentration of 1.3 to 2.2 mmol/L (~23-40mg/dL) can be used as a diagnostic aid for a septic joint6. However, glucose concentration gradients should not be relied upon to make a diagnosis of septic arthritis because insufficient studies have been performed in animals with high leukocyte and neutrophil counts due to non-infectious causes (likely because these are uncommon in horses).
The most common bacteria reported in septic joints in foals are gram negative bacteria from the family Enterobacteriaceae, Escherichia coli is the most common isolate from young foals with hematogenous spread being the most common route of infection4. Nevertheless, one retrospective study from our institution found that gram positive bacterial infections (due to Streptococcus or Staphylococcus species) were as common as gram negative bacterial infections (mostly E. coli) in foals with septic arthritis2. Gram positive bacterial infections are more frequently associated with arthritis in older horses and the route of infection is more commonly iatrogenic, related to direct trauma to the joint capsule or a nearby joint soft tissue trauma10. Gram positive bacterial infections in foals are associated with a lower morbidity than gram negative bacterial infections2, although this observation may change with the evolution of multidrug resistant bacterial cocci. Foals that have positive cultures for multiple pathogens have a more guarded prognosis for survival2.
In this case, the offending pathogen was S. zoepidemicus, a gram positive β-hemolytic bacterium, which has been reported to be one of the common gram negative bacteria causing septic joints in foals2,4,10 and adult horses3. S. zooepidemicus is part of the normal flora of the oral cavity, pharynx and respiratory tract in horses and is thought to cause disease as an opportunistic pathogen10. Given the history of this foal potentially sustaining trauma two days prior to presentation and the fact that the foal had a lower than desired for IgG concentration upon presentation, reduced immunity could have predisposed the foal to an opportunistic infection with S. zooepidemicus in the hip joint. The reduced immunity could have been an acquired condition, because the foal was reported to have adequate IgG concentrations after parturition (value not provided). Foals are born immunocompetent and low concentrations of immunoglobulins are detectable at birth, however, evidence of a primary immune response to external stimuli is not detectable until about two weeks of age11. It is generally believed that foals of this age should ideally have IgG concentrations at or above 800 mg/dL to be able to adequately fight off an infectious agent.
Follow up
The foal was hospitalized for one week and was discharged with no further complications. Because bacterial sepsis is usually a presumptive diagnosis based on high total nucleated cell and neutrophil counts, finding bacteria on cytologic assessment made this case rewarding, especially when we can give a preliminary definitive diagnosis of cause (and presumptive speciation) with an easy and practical test such as joint fluid cytologic analysis.
References
- Edward A. Mahaffey. Diagnostic Cytology and hematology of the Horse. In: Schrefer JA, editor. Diagnostic Cytology and Hematology of the Horse. 2nd ed. St. Louis, Missousi: Mosby; 2002. p. 163–70.
- Vos NJ, Ducharme NG. Analysis of factors influencing prognosis in foals with septic arthritis. Ir Vet J [Internet]. 2008;61(2):102–6.
- Schneider RK, Bramlage LR, Moore RM, Mecklenburg LM, Kohn CW, Gabel a a. A retrospective study of 192 horses affected with septic arthritis/tenosynovitis. Equine Vet J. 1992;24(6):436–42.
- Hepworth-warren KL, Wong DM, Fulkerson C V, Wang C, Sun Y. Bacterial isolates, antimicrobial susceptibility patterns, and factors associated with infection and outcome in foals with septic arthritis:83 cases (1998 – 2013). J Am Vet Med Assoc. 2015;246(7):785–93.
- Newquist JM, Baxter GM. Evaluation of plasma fibrinogen concentration as an indicator of physeal or epiphyseal osteomyelitis in foals: 17 cases (2002-2007). J Am Vet Med Assoc. 2009;235:415–9.
- Morton AJ. Diagnosis and treatment of septic arthritis. Vet Clin North Am – Equine Pract. 2005;21(3):627–49.
- Hardy J. Etiology, Diagnosis, and Treatment of Septic Arthritis, Osteitis, and Osteomyelitis in Foals. Clin Tech Equine Pract. 2006;5(4):309–17.
- Erol E, Locke SJ, Donahoe JK, Mackin MA, Carter CN. Beta-hemolytic Streptococcus spp. from horses: a retrospective study (2000-2010). J Vet Diagnostic Invest [Internet]. 2012;24(1):142–7.
- Madison JB, Sommer M, Spencer PA. Relations among synovial membrane histopathologic findings, synovial fluid cytologic findings, and bacterial culture results in horses with suspected infectious arthritis: 64 cases (1979–1987). J Am Vet Med Assoc 1991;198:1655–1661.
- Annear MJ, Furr MO, White NA. Septic arthritis in foals. Equine Vet Educ. 2011;23(8):422–311.
- Liepman RS, Dembek KA, Slovis NM, Reed SM, Toribio RE. Validation of IgG cut-off values and their association with survival in neonatal foals. Equine Vet J. 2015;47:526–30
Authored by: José Daniel Cruz Otero (Clinical Pathology Resident) and T. Stokol
