Whole blood is collected from a suitable donor animal for blood transfusion purposes into a blood bag containing citrate phosphate dextrose as the anticoagulant. Once the blood has been collected, it can be stored at 4°C and used in its natural state or can be converted into a variety of components. These blood components are packed red blood cells, platelet-rich plasma, platelet concentrates, fresh plasma, fresh frozen plasma, frozen plasma, cryoprecipitate and cryosupernant. Double, triple and quadruple blood bags (a single whole blood collection bag with various satellite bags) are used for producing and separating components within a sterile closed environment. Blood component production is highly desirable for these reasons:
- Components maximize the yield of products from a single blood donation.
- Ability to use the optimal products (in high concentrations) for specific diseases.
- Minimize exposure to unnecessary foreign material in recipients, particularly red blood cell antigens, which can incite transfusion reactions.
- Increased efficacy of treatment through providing only the factors that are required in the animal, e.g. cryoprecipitate for treatment of von Willebrand disease or Hemophilia A (cryoprecipitate is enriched in von Willebrand factor and Factor VIII, which are deficient in these two diseases, respectively).
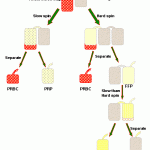
The most commonly available blood components are whole blood, packed red blood cells, fresh frozen plasma and cryoprecipitate. Production of these components requires a specific type of refrigerated centrifuge (bucket centrifuge) which can fit the large transfusion bags and sediment cells from plasma. Click on the thumbnail to the right to see a visual representation of how these products are produced.
Whole blood
This is blood collected directly from a donor animal into a blood transfusion bag containing citrate-phosphate-dextrose with (CPDA-1) or without adenine (CPD) as an anticoagulant. For dogs, a 500 mL transfusion bag is used (which contains approximately 63 mL of anticoagulant, obtaining approximately 450 mL blood from the donor dog). For cats, anticoagulant is usually placed in a 30 or 60 mL syringe (in the appropriate anticoagulant to blood volume ratio), although pediatric transfusion bags can be used. Production of components from whole blood (with complete sterility) is only achievable if blood is collected into transfusion bags. Whole blood can be used immediately or stored at 4°C for future transfusion or separation into blood components.
Additives have been developed that can be added to the whole blood in a sterile fashion (they are attached to the original transfusion bag as a satellite bag). These are used to optimize red blood cell storage viability. These additives usually contain dextrose and adenine, for red blood cell energy metabolism, and mannitol, which decreases red blood cell lysis. Several additives have been evaluated with canine red blood cells. Both Nutricel® and Adsol® increase the post-transfusion viability of stored canine red cells to 35 and 37 days, respectively. Separation of whole blood into components ideally requires a refrigerated centrifuge with rotor heads large enough to hold a 500 mL blood bag – these are generally bucket centrifuges and refrigeration of the centrifuge is required to produce cold-dependent components such as cryoprecipitate.
For species in which red blood cells settle rapidly with gravity alone (horses), compression devices can be used to obtain red blood cell-depleted plasma and red blood cells. The red blood cells are allowed to settle in the bag at room temperature (takes a minimum of 20 minutes), then the bag is placed in a gentle compression device which squeezes the supernatant plasma into a separate transfusion bag. The plasma contains some leukocytes, platelets (which can cause febrile transfusion reactions) and some red blood cells (which will sensitize the recipient to foreign red blood cell antigens). This procedure is not commonly used due to the less than clean separation of red blood cells from plasma, but is an option when packed red blood cells are not available and the donor’s plasma contains antibodies which will destroy the recipient’s red blood cells (i.e. an incompatible minor cross-match reaction).
RBC components
Packed red blood cells

These are a concentrated source of red blood cells that remain in a small amount of plasma once supernatant plasma is removed after centrifugation of the blood transfusion bag containing whole blood. Packed cells are an excellent source of red blood cells for anemic animals that need the additional oxygen-carrying capacity these cells provide. Packed red blood cells can be used immediately or stored at 4°C. They need dilution in sterile isotonic solution prior to infusion (due to the high hematocrit, packed cells without dilution are very thick and flow sluggishly through infusion lines). The current standard for red blood ell storage is that there should be about 75% post-transfusion viability after storage, that is at least 75% of the transfused red blood cells must remain in the recipient’s circulation 24 hours after transfusion. The post-transfusion viability of canine red blood cells collected into CPDA-1 is 20 days. Changes that do occur in packed red blood cells with storage over 28 days include a decrease in pH (mean 7 to 6.4 units), likely due to increased lactate concentration (mean 2.7 to 19.7 mmol/L), increased potassium concentrations (mean 2.7 to 6.7 mEq/L; attributed to defective sodium-potassium pumps in cells) and increased hemolysis (mean of 0.2% to 0.7%, with higher hemolysis seen in individual units, up to 2%). The stored red blood cells were administered to dogs without overt complications (Rodrigues et al 2020).
Platelet components
Platelet-rich plasma
Platelet-rich plasma is produced by separating plasma from red blood cells (within 6 hours of whole blood collection) using a slow spin to prevent pelleting of the platelets. Platelet-rich plasma is useful for the treatment of disorders of platelet number and function but must be infused within 8 to 12 hours (maintained at room temperature) due to platelet instability (which limits its use).
Platelet concentrates
Platelet concentrates are produced by separating most of the plasma from platelet-rich plasma. Platelet concentrates are used similarly to platelet-rich plasma.
Plasma components
Platelet-poor plasma: Fresh, fresh-frozen, frozen
Platelet-poor plasma is produced by separating plasma from red blood cells using a high spin to pellet platelets with the red cells. This is the starting point for production of most blood components. Separation of platelets is desirable because they provide an additional source of foreign antigens and micro-aggregation can cause transfusion reactions. Approximately 200 to 400 mL of plasma are obtained from each whole blood transfusion bag. One unit of plasma is defined as that obtained from a single whole blood transfusion bag.
- Fresh plasma: This is platelet-poor plasma that is separated from red cells and infused within 6 hours of blood collection (with the blood bag being maintained at 4°C until separation).
- Fresh frozen plasma (FFP): This is platelet-poor plasma that is separated from red cells within 6 hours of blood collection and frozen in a dedicated freezer (at or below -18°C).
- Fresh frozen plasma and fresh plasma contain all coagulation factors and plasma proteins (such as albumin).
- Stable for 1 year if maintained in a dedicated freezer (one that does not undergo freeze-thaw cycles like household freezers) at or below -18°C
- One unit of FFP is defined as that obtained from a single whole blood transfusion bag (approximately 250 mL).
- Frozen plasma: Frozen plasma lacks certain coagulation factors which are quite unstable, including factor VIII, von Willebrand factor and Factor V. It is a source of plasma proteins and inhibitors. Frozen plasma is stable for 1 or 7 years if stored at or below -18°C or -65°C in a dedicated freezer, respectively. It can come from several sources:
- Plasma that is separated and frozen longer than 6 hours after whole blood collection.
- Fresh plasma that is not used within 6 hours of collection and then frozen.
- Fresh frozen plasma maintained for longer than 1 year in a dedicated freezer at or below -65°C (stable for an additional 6 years).
Cryoprecipitate (CPP) and cryosupernatant (cryosuper)
These products are produced by slow thawing of fresh frozen plasma at 4°C. Cryoprecipitable proteins (von Willebrand factor, factor VIII, fibronectin, fibrinogen) precipitate at this temperature and can be concentrated in a very small amount of remaining plasma (approximately 1/10 of the starting volume of FFP, or about 20 to 30 mL).

- Cryoprecipitate: This is produced by slow-thawing (at 4°C) fresh frozen plasma, followed by centrifugation at 4°C and removal of most of the supernatant plasma.
- Concentrated source of von Willebrand factor, fibrinogen, Factor VIII and fibronectin.
- Cryoprecipitate is stable for 1 year from the date of collection of the whole blood for transfusion purposes (not the date of preparation of the product) if maintained at or below -20°C in a dedicated (not frost free) freezer.
- One unit of cryoprecipitate is defined as that obtained from a single FFP bag (approximately 250 mL plasma).
- Cryosupernatant (Cryosuper): This is the remaining plasma after removal of the pelleted cryoprecipitate and is considered equivalent to frozen plasma (see above).
- Source of all coagulation and plasma proteins, except for Factor VIII, fibrinogen, von Willebrand factor and fibronectin.
- Cryosupernatant is stable for 1 years if stored in a dedicated freezer at or below -18°C and 7 years if stored at -65°C or lower.
