Physiology
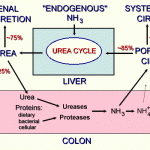
Ammonia is produced from dietary amino acids and by catabolism of amino acids, amines, nucleic acids, glutamine and glutamate (nitrogenous wastes) in peripheral tissues (especially skeletal muscle). Gastrointestinal micro-organisms (primarily coliforms and anaerobes in the colon and cecum) convert dietary amino acids and urea into ammonia in the gastrointestinal system. The ammonia is absorbed into the portal circulation, taken up by the liver and converted in the liver, via the urea cycle, into urea. Urea is then excreted into the gastrointestinal system (producing a futile cycle) and into the urine.
Of the total ammonia produced, 80-90% is shunted into the urea cycle, with the remaining 10-20% metabolised by peripheral tissues, including the kidney, heart, and brain.
Methods
The method used by Cornell University to measure ammonia is given below.
Reaction type
Enzymatic end-point, with glutamate dehydrogenase (GLDH).
Procedure
Glutamate dehydrogenase (GLDH) catalyzes the reductive amination of 2-oxoglutarate with NH4+ and NADPH to form L-glutamate and NADP+.
The declining rate of NADPH is measured photometrically and is proportional to the ammonia concentration.
NH4++ 2-oxoglutarate + NADPH GLDH > L-glutamate + NADP+ + H2O
Units of measurement
Ammonia levels are measured in μg/dL (conventional units) and μmol/L (SI units). The conversion formula is as follows:
μg/dL x 0.587 = μmol/L
Sample considerations
Sample type
Plasma, cerebrospinal fluid (CSF), aqueous humor, vitreous humor
Anticoagulant
Heparin, EDTA
Stability
Measurement of ammonia is problematic as it is very unstable. Arterial blood samples are preferable to venous blood samples as results are more consistent. Heparinized or EDTA-anticoagulated plasma samples are preferable to serum. Due to the instability of ammonia and leakage of ammonia from erythrocytes, whole blood samples should be kept on ice, then separated within 15 minutes of collection. Separated samples to be assayed for ammonia as soon as possible after sample collection and maintained at 4°C (or on ice) until assayed (stable for a maximum of 3 hours under these conditions). Alternatively, samples can be frozen and stored frozen (and shipped as such) until analysis. Studies in horses indicate that ammonia is stable for 21 days under frozen storage.
The sample must be separated from cells as soon as possible as leakage of ammonia from erythrocytes occurs within 30 minutes, resulting in falsely high values. This increase is due to ammonia generation from labile proteins and amino acids (e.g. glutamine), production from both WBC and RBCs. Furthermore, any ammonia in the environment (air, water supply) can contribute to the ammonia in the patient sample. Decreased values can occur if the blood tube is not completely filled nor stoppered, through loss of ammonia gas.
Ammonia assays, therefore are not routinely performed. Control samples (from a clinically healthy animal) should always be run in conjunction with patient samples, to ensure that sample collection and handling are not responsible for elevations in ammonia.
For post-mortem analysis of ammonia in cerebrospinal fluid or ocular fluids for diagnosis of ammonia toxicity, samples should be collected similarly from control animals to obtain guidelines on expected results. In vitreous humor from human patients, ammonia will increase linearly over time after death and can be used to estimate time of death (Henry and Smith 1980). Ammonia also increases with time in aqueous humor samples collected from cattle immediately after death and maintained at 4ºC for 24 hours before analysis (Fitzgerald et al 2006).
Summary of Collection Guidelines
- Collect samples into EDTA (purple top, preferred) or heparin (green top).
- Keep sample on ice.
- Centrifuge and separate plasma within 15 minutes (ideally) of collection.
- Keep separated plasma on ice or refrigerated and assay within 3 hours.
- If longer delay until analysis anticipated, freeze rapidly and store in a frost-free (dedicated) freezer at -20ºC.
- Ship on dry ice and mark package ” AMMONIA measurement, KEEP FROZEN”. The sample MUST NOT thaw during transit so ensure there is sufficient dry ice to maintain frozen samples.
- Contact our laboratory ahead of time to notify of shipment and estimated time of arrival, so we can run controls and calibrate our instrumentation in preparation for rapid analysis (within 1 hour if kept cold) once thawed.
- Result accuracy cannot be assured for samples that are arrive thawed due to the instability of ammonia.
Post-mortem measurement
Measurement of ammonia concentrations in cerebrospinal fluid (CSF) and aqueous or vitreous humor post-mortem should be undertaken with caution because ammonia concentrations are expected to increase after death (Henry and Smith 1980). However, ammonia concentrations have been measured within 1 hour of collection (kept on ice) in CSF and aqueous humor after a 10 hour post-mortem interval in a horse suffering from hyperammonemia secondary to intestinal disease (the carcass was kept refrigerated during these 10 hours). Results in the horse were far higher than in samples collected from 6 other horses (CSF: 1566 umol/L in the affected animal versus 370-530 umol/L in the 6 unaffected horses; aqueous humor: 1018 umol/L in the affected horse versus 70-483 umol/L in the 6 other horses), whose carcasses were handled similarly (Gilliam et al 2007). However, ammonia concentrations would be expected to rise much higher than that seen in the 6 horses if carcasses were not maintained under controlled refrigerated conditions or if there was red or white blood cell contamination of the samples.
Interferences
- Lipemia/Turbidity: Severe lipemia may increase concentrations (>170 turbidity index, per product information sheet)
- Hemolysis: May increase with severe hemolysis (>50 hemolysis index per product information sheet)
- Icterus: Severe icterus may increase concentrations (>60 icteric index per product information sheet)
Test interpretation
Only increased ammonia concentrations are diagnostically useful, indicating hepatic dysfunction or identifying hyperammonemia (regardless of a cause) of neurologic symptoms. In general, baseline ammonia concentrations are measured in animals with suspected abnormal portal blood flow or hepatic dysfunction. Ammonia is also measured in animals showing neurologic symptoms. Ammonia tolerance tests can be performed to increase sensitivity of the test to abnormal portal blood flow. Just like a post-prandial bile acid concentration, the ammonia tolerance test involves giving the animal ammonia as a challenge to see how the liver handles the excess ammonia. However, this test can precipitate clinical signs of encephalopathy so it is seldom used.
Increased ammonia concentration (hyperammonemia)
Ammonia is toxic to the CNS and is one of the causes of hepatic encephalopathy (but not the sole cause) with high values predicting clinical signs of encephalopathy in dogs with congenital shunts (Tivers et al 2014).
- Artifact: Delayed sample submission, delayed plasma separation and hemolysis. This is the most common cause for an increased ammonia concentration.
- Physiologic: Physiological increases in ammonia occur after high protein meals and strenuous exercise.
- Pathophysiologic
- Decreased uptake of ammonia:
- Abnormalities in hepatic portal blood flow: Ammonia can increase in blood with congenital or acquired shunts. In one study, fasting ammonia was less sensitive than fasting bile acid concentrations in symptomatic dogs with congenital shunts (van Straten et al 2015). High concentrations of ammonia, with high fasting bile acid concentrations, were specific for shunts in the latter study. Ammonia tolerance tests may be performed in animals with suspected shunts and may be slightly more sensitive than fasting bile acids (van Straten et al 2015).
- Hepatic dysfunction: When >60-70% of hepatic function is lost, ammonia concentrations can increase due to inadequate uptake and conversion to urea. With both abnormal portal blood flow and hepatic dysfunction, defects in extraction of ammonia or conversion to urea, can result in the following
- Increased plasma ammonia can lead to increased renal excretion and formation of ammonium biurate crystals in urine (which can be a diagnostic marker of hepatic dysfunction or abnormal portal flow in some animals.
- Decreased urea concentrations in blood: This can result decreased urine concentrating ability (inappropriately concentrated urine in azotemic animals or even isosthenuria since urea is a major contributor to the medullary concentration gradient).
- Decreased conversion to urea:
- Hepatic dysfunction/abnormal blood flow: See above. Some dogs with congenital shunts have evidence of downregulation of urea cycle intermediate protein genes and abnormal zonal distribution of these proteins, which could affect ammonia metabolism to urea (van Straten et al 2014).
- Inherited disorders in the urea cycle: These have been reported rarely in Irish Wolfhounds (Zandfliet and Rothuizen 2007) and cats (Washizu et al 2004). Bile acid concentrations are expected to be normal in these conditions. Persistent hyperammonemia has been reported in two related Morgan weanlings and was thought to be due to defective mitochondrial transport of ornithine, a required intermediate in the urea cycle (McCornico et al 1997).
- Lack of or decreased availability of urea cycle intermediates, e.g. arginine deficiency in cats (Morris and Rogers 1978).
- Organic acidemias: Methylmalonic acidemia secondary to acquired or inherited cobalamin deficiency is associated with hyperammonemia in dogs (e.g. Beagles and Border collies with an inherited defect in cubilin [Fyfe et al 2014]) and cats (Vaden et al 1992). The reason for the high ammonia with this disorder is unclear, but methylmalonic acid may disrupt activity of urea cycle enzymes.
- Increased production of ammonia:
- Overgrowth of urease-producing bacteria in the gastrointestinal tract:
- Rumen: Urea and soybean meal toxicosis in ruminants is also associated with high ammonia, from urease producing bacteria in the rumen (Ortolani et al 2000, Raboisson et al 2012).
- Large intestine: Overgrowth of these bacteria in the large intestine of some horses with diarrhea can produce a hyperammonemia with accompanying neurologic signs (head pressing, blindness and maniacal behavior), e.g. colitis (Dunkel et al 2011), necrotizing enteritis associated with coronavirus infection (Giannitti et al 2015). This is likely the cause of an “idiopathic” hyperammonia in a horse with diarrhea (Gilliam et al 2007)
- Toxicity: Anhydrous ammonia (Fitzgerald et al 2006) or urea toxicity in cattle.
- Overgrowth of urease-producing bacteria in the gastrointestinal tract:
- Decreased uptake of ammonia:
