Inherited coagulopathies arise from mutations within genes responsible for synthesis or processing of active coagulation factors. In animals, deficiencies of intrinsic pathway factors (Factor XII, Factor XI, Factor IX and Factor VIII) are the most common. Some of these diseases are sex-linked (the gene is on the X chromosome) and usually occur in males. The degree of factor deficiency and its role in coagulation will determine the bleeding tendency associated with each trait.
Factor II deficiency
Inherited disorders of Factor II (prothrombin) are extremely rare and have only been described in the English Cocker Spaniel and Boxer breed. In humans and Boxers, the defect has an autosomal recessive inheritance pattern. The disorder in the Boxer was one of dysprothrombinemia (non-functional prothrombin), rather than absolute deficiency of the protein since immunoassay measurements of prothrombin concentrations were normal. Whether the deficient activity of prothrombin in the Cocker Spaniel was due to lack of prothrombin or dysfunctional prothrombin was not determined.
Signs in affected puppies include epistaxis and gingival bleeding. Bleeding episodes tend to be milder with maturation; affected adult dogs have a tendency to bruise and/or develop dermatitis. Coagulation studies in untreated puppies were characterized by a prolonged ACT, APTT, and PT. Transfusion of fresh whole blood can successfully arrest bleeding episodes for up to three days. Fresh or fresh-frozen plasma transfusion is preferred if the animal does not require red blood cells..
Factor VII deficiency
Factor VII (proconvertin) deficiency has been recognized in colony-bred mongrels, Beagles, Miniature Schnauzers, Alaskan Malamutes, Boxers, and Bulldogs. The inheritance pattern in Beagles is autosomal with incomplete dominance. In colony-bred mongrels, the disorder is autosomal recessive.
Factor VII deficiency is not usually accompanied by detectable bleeding. Affected individuals may experience bruising or prolonged bleeding following surgery, postpartum hemorrhage and hematoma formation. Usually, the condition is discovered incidentally during screening tests for blood clotting ability. It is characterized by a prolonged PT and a normal APTT or ACT.
Factor VIII deficiency (Hemophilia A)
Factor VIII (antihemophilic factor) deficiency, or hemophilia A, is the most common inherited coagulation factor deficiency in dogs and cats. It has also been recognized in several breeds of horses (including Arabs, Standardbreds, Thoroughbreds, and Quarter horses), and in Hereford cattle.
The disease arises from a spontaneous mutation in the factor VIII gene which lies on the X-chromosome. Due to the X-linked recessive mode of inheritance, males are usually affected (by maternal transmission), while females are asymptomatic carriers. The birth of affected female progeny requires the mating of an affected male to a carrier female, a possibility in closely inbred families or mildly affected male hemophiliacs.
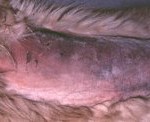
The severity of clinical signs are dependent on the degree of deficiency in factor VIII (see table below). Severely affected dogs show the most severe bleeding symptoms, with moderately and mildly affected dogs showing less severe signs. Spontaneous hematomas in subcutaneous tissues, hemarthrosis and hemorrhagic body cavity effusions are common manifestations in affected dogs, particularly those with severe deficiencies. Excessive hemorrhage is also observed with traumatic events (e.g. shedding of deciduous teeth, elective surgery) in dogs with hemophilia A regardless of severity. Bleeding after venipuncture or trimming of nails was only recognized in dogs with severe deficiency in a review of 39 cases (Aslanian et al 2014). Dogs with milder deficiency may demonstrate subtle symptoms which may go unrecognized if the dog is not subject to surgery or trauma. A recurrent shifting-leg lameness may be the only manifestation observed (due to mild hemarthrosis) and is certainly not specific for a bleeding disorder (in fact, an infectious or immune-mediated cause would be higher on the differential diagnostic list). Some dogs may even reach breeding age, thus disseminating the genetic defect widely. Indeed, the majority of German Shepherd dogs with hemophilia A in most countries (including the USA and Australia) are thought to have descended from one male dog from Germany, who was a highly desirable and prolific sire.
Factor VIII deficiency in cats can be associated with spontaneous hematomas, but presumably due to their light weight and agility, affected cats often do not experience prolonged hemorrhage except following trauma or surgery. A similar presentation may also be observed in small breed dogs. In horses, affected colts usually die at or after birth from severe hemorrhage. Any mare, with a history of several male foals with hemorrhage-related perinatal death, should be checked for hemophilia A.
Factor VIII deficiency usually causes a prolonged APTT and ACT (although the ACT may be normal in patients with mild or moderate hemophilia A). Specific diagnosis (and differentiation from hemophilia B or factor IX deficiency) requires determination of specific factor activity in coagulation assays. Factor VIII-coagulant (FVIII:C) activity ≤ 20% is consistent with a diagnosis of hemophilia A, although in most dogs, factor VIII:C activity is < 6% (moderate to severe deficiency). Carriers of hemophilia A have reduced levels of factor VIII (40 to 60%) compared to normal dogs (>60% activity), while von Willebrand factor antigen levels are normal or greater than normal (producing vWf:Ag/Factor VIII:C ratios > 2, however this ratio should not be relied upon for carrier detection). Identification of carriers should be done with caution in animals < 6 months of age (as young animals have lower factor VIII:C activities than adults). Carriers usually have a normal APTT and ACT. Currently, the genetic defects in animals with hemophilia A have not been identified, precluding any genetic tests for carrier detection.
|
Severity
|
Factor VIII: Coagulant activity | ||
|---|---|---|---|
|
Mild
|
6 – 20 % | ||
|
Moderate
|
2 – 5 % | ||
|
Severe
|
< 2 % |
Treatment for hemophilia A requires repeated transfusions of fresh plasma, fresh-frozen plasma or cryoprecipitate 2 to 3 times a day until bleeding is under control. Similar to patients with von Willebrand disease, plasma and cryoprecipitate transfusions are preferred due to the possible sensitization of the animal to red blood cell antigens. Cryoprecipitate is generally more effective than fresh or fresh-frozen plasma in the treatment of hemophilia A. A study of 39 dogs with hemophilia A demonstrated that the prognosis is fair in affected dogs, even those that are of large breeds. Even though affected dogs suffer from recurrent bouts of hemorrhage, they can usually be successfully managed with transfusion therapy (Aslanian et al 2014).
View the Hemophilia A fact sheet by the Comparative Coagulation Laboratory of the Animal Health Diagnostic Center at Cornell University.
Factor IX deficiency (Hemophilia B)
Factor IX (Christmas factor) deficiency, or hemophilia B, is an X chromosome-linked hemorrhagic disorder identical in presentation to Factor VIII deficiency (hemophilia A). As with Factor VIII, the disorder primarily affects males, except in closely inbred families where a homozygous female can result from the mating of an affected male to a carrier female. Although not as common as hemophilia A, hemophilia B has been described in one mixed-breed dog and in many pedrigrees, including Labrador Retrievers and German Shepherd dogs. It has been recognized in one family of British Shorthair cats and Siamese-cross cats.
In cases of severe Factor IX deficiency (activity < 1% of normal), puppies or kittens may die at or immediately following birth. Excessive bleeding from the umbilical cord or tail and feet at the time of tail docking and dewclaw removal are common signs. Hemarthrosis, gingival bleeding during tooth eruption, and spontaneous hematoma formation are other typical manifestations.

Individuals with Factor IX levels between 5 and 10% may not be recognized as being hemophiliacs unless challenged with trauma or surgery. Small dogs and cats also often go unrecognized as having a bleeding disorder despite the presence of very low levels of Factor IX, presumably due to their light weight and usually protected environments. Most affected individuals will have a prolonged APTT (and perhaps ACT). Carriers of hemophilia B usually have Factor IX levels between 40 and 60% and cannot be detected on routine coagulation screening tests. Identification of a carrier state in animals < 6 months of age by factor activity evaluation should be conducted caution since young animals normally have lower activity than adults. Age-matched controls are important but may not eliminate the necessity of re-evaluating the individual at 8 to 12 months of age. The genetic defect in some purebreeds (e.g. Lhasa Apso) has been identified and PCR-based genetic techniques may be used to identify the mutation in suspected carriers.
Transfusion of fresh plasma, fresh-frozen plasma or cryosupernatant is the recommended treatment for acute bleeding episodes. Although external hemorrhage is fairly easy to discern, affected individuals often bleed internally into the thorax or abdomen, between fascial planes separating muscle groups, or into the brain. Often, these bleeding episodes are not recognized until a crisis exists. Unchecked hemorrhage can result in muscle necrosis, paralysis, seizures, and/or hypovolemic shock.
View the Hemophilia B fact sheet by the Comparative Coagulation Laboratory of the Animal Health Diagnostic Center at Cornell University.
Factor X deficiency
Factor X (Stuart factor) deficiency is a rare hemorrhagic disorder that has only been described in a family of American Cocker Spaniels, in a female Jack Russell terrier and a male Domestic Shorthair cat. The inheritance pattern in the American Cocker Spaniels is autosomal dominant with variable penetrance. In American Cocker Spaniels, there is a high neonatal mortality rate. Affected individuals show moderate to severe hemorrhagic tendencies. The female Jack Russell terrier had recurrent episodes of spontaneous and surgical (post-taildocking and post-ovariohysterectomy) hemorrhage. Individuals with Factor X levels < 30% usually have a prolonged PT, APTT and perhaps ACT. The cat presented with excessive hemorrhage after venipuncture and declawing. The PT and APTT were prolonged, with normal fibrinogen, vWf:Ag and platelet count. Factor X activity was < 2%. Treatment to control bleeding requires the intravenous infusion of fresh or fresh-frozen plasma or cryosupernatant.
Factor XI deficiency (Hemophilia C)
Hemophilia C is rare and has only been described in the Springer Spaniel, Great Pyrenees, Weimaraner, and Kerry Blue terrier breeds. Dogs with severe Factor XI (plasma thromboplastin antecedent) deficiency characteristically have minimal bleeding until subjected to trauma or surgery, at which time major bleeding occurs. In some cases, bleeding may be delayed for up to 4 days after a surgical procedure. The mode of inheritance is autosomal (therefore males and females can be affected); it is not clear whether the gene is dominant or recessive. Affected individuals with Factor XI levels < 30 to 40% will usually have a prolonged APTT and perhaps ACT. Bleeding episodes can be controlled by the intravenous infusion of fresh plasma, fresh-frozen plasma or cryosupernatant.
In human patients with hemophilia C, the hemorrhage has been mostly attributed to excessive fibrinolysis. With Factor XI deficiency, coagulation is not amplified by thrombin (which usually amplifies the pathway through activation of Factor XI). The small amount of thrombin generated by the tissue factor (extrinsic) pathway is insufficient to activate a fibrinolytic inhibitor, tissue factor pathway inhibitor (TAFI). The lack of TAFI in patients with Factor XI deficiency thus promotes fibrinolysis of the clots that are initially generated, explaining why hemorrhage is sometimes delayed for several days after a surgical procedurer.
Factor XI deficiency has been recognized in Holstein cattle and is inherited as an autosomal recessive trait. Similar to dogs and humans, cattle rarely show spontaneous bleeding; rather bleeding appears to be induced by trauma or surgery (such as dehorning).
Factor XII deficiency
Factor XII (Hageman factor) deficiency has been reported in cats, in individual dogs of various breeds, and a family of Miniature Poodles. The genetic defect causing factor XII deficiency has been identified in cats. It is due to a single base deletion in exon 11 of the factor XII gene, resulting in a premature truncation of the protein. The mutation was seen in all 8 tested cats but not 4 healthy controls (Bender et al 2015). In the Miniature Poodle, Factor XII deficiency occurred with von Willebrand disease; the combination did not seem to exacerbate bleeding (Randolph et al 1986). Although Factor XII deficiency is not associated with apparent bleeding, affected individuals may be predisposed to infection and/or thrombosis. This may be related to the central role of Factor XII in the activation of inflammatory mediators, such as bradykinin, and the fibrinolytic pathway (Woodruff et al 2007). Factor XII is normally lacking in the plasma of birds, marine mammals, and reptiles (Robinson et al 1969, Saito et al 1974); the APTT is not a useful diagnostic test in these species.
Factor XIII deficiency
Factor XIII is a transglutaminase enzyme that crosslinks the γ-chains of two D-domains of fibrin (creating the neo-epitope, D-dimer in the process and forming a stabilized longitudinal fibrin polymer) and the α-chains of two adjacent fibrin polymers, forming a laterally (cross-sectionally) stabilized fibrin network. It is activated by thrombin (factor IIa) in the presence of calcium and fibrin. There have been two reports of factor XIII deficiency in dogs. A presumptive diagnosis of congenital deficiency of factor XIII was made in a Toy Poodle cross with recurrent traumatic- or surgical-induced hemorrhage (subcutaneous hematoma, post-surgical hemorrhage from incision sites, etc). The dog had normal coagulation screening test results (PT, APTT) and vWf:Ag results. A thrombopathia was ruled down on the basis of normal closure time with the platelet function analyzer and the collagen/ADP cartridge. On thromboelastometry (Rotem® analyzer), the clot formation time was very prolonged (ellagic acid-activated) or immeasurable (tissue factor-activated) with a low maximum clot firmness (MCF). A FXIII solubility assay was performed at a human hospital and was abnormal. With this assay, plasma is induced to clot in the presence of thrombin and calcium, then a solution is added to achieve clot lysis (e.g. 5M urea) over 24 hours at 37ºC. More rapid clot lysis (<24 hours) occurs in the presence of a weak clot, such as that formed in the absence of FXIII (<1% deficiency). Post diagnosis, the dog continued to suffer from recurrent bleeds. In human patients, the treatment of choice is cryoprecipitate or fresh frozen plasma (Kong et al 2014). The second case was a 4 month old Coonhound, which suffered from multiple episodes of bleeding, including post-surgical bleeding (from incision sites after an oral biopsy, post-splenectomy), hemoperitoneum from apparent trauma, presumptive hemoarthrosis (shifting leg lameness) and bleeding from the mouth. Hemorrhage was successfully treated with combinations of plasma products (fresh frozen plasma, cryoprecipitate) and fibrinolytic inhibitors. Hemostasis testing revealed mostly normal screening coagulation times and von Willebrand factor antigen. Viscoelastic testing showed reduced clot firmness and increased fibrinolysis. There was enhanced fibrinolysis in high concentrations of urea (5 M), suggestive of a factor XIII deficiency, which was confirmed using a chromogenic assay for factor XIII activity in humans. A deletion-insertion mutation was found in the A1 unit of the factor XIII tetramer, which resulted in a frameshift and a premature stop codon. This mutation would prevent the protein from being translated and explain the deficiency (Pieples et al 2026).
Prekallikrein deficiency
Prekallikrein deficiency has been reported in a family of Belgian horses, a family of miniature horses and in two dogs. In the family of Belgian horses, one horse bled excessively after castration was performed. One of the canine reports was in a young Chinese Shar Pei dog, with combined Factor XII and partial prekallikrein deficiency. This combined deficiency was thought to be the cause of recurrent gastrointestinal hemorrhage in the patient (although this would be unusual due to the role [lack of] of both these molecules in coagulation).
Prekallikrein (or Fletcher factor) is a proenzyme that circulates in plasma in a 1:1 complex with high-molecular weight kininogen (HWMK). Prekallikrein is converted to kallikrein by Factor XIIa. Once formed, kallikrein enhances the activation of Factor XII and (with Factor XIIa) converts HWMK into bradykinin, which is an important mediator of vasodilation, inflammation and fibrinolysis (by stimulating the release of tissue plasminogen activator from endothelial cells).
A deficiency of prekallikrein does not cause a clinically apparent coagulopathy, but does result in a prolonged APTT (and possibly ACT). Affected animals are usually detected in the course of coagulation testing either for bleeding disorders caused by other conditions, screening prior to surgery or initiation of research protocols. In prekallikrein deficiency, extended incubation of the reagents with the patient plasma prior to addition of calcium chloride will shorten the prolonged APTT (as prekallikrein enhances factor XII activation, factor XII activation can proceed in the presence of a deficiency of prekallikrein as long as there is extended contact with the thromboplastin reagent). The prolonged APTT should also correct with addition of pooled plasma from normal animals to the patient plasma.
Deficient or dysfunctional fibrinogen
Congenital afibrinogenemia has been reported in one family of Saanen dairy goats and has not been described in dogs or cats. Hypofibrinogenemia has been reported in the St. Bernard and Vizsla breeds. Bleeding was severe, with a prolonged ACT, APTT and PT, and low fibrinogen (measured by the thrombin clot time) and fibrinogen antigen.
Dysfibrinogenemia (abnormal structure with defective function) has been recognized in one inbred family of Russian wolfhounds. Coagulation screening test results included a prolonged ACT, APTT, and PT with low fibrinogen measured by thrombin clot time. Fibrinogen antigen could be detected by precipitation with antibody against fibrinogen. Affected animals experienced mild bleeding manifested by lameness and epistaxis; challenge with surgery or trauma resulted in life-threatening bleeding.
Dysfibrinogenemia or afibrinogenemia has also been reported in a Border Leicester lamb, with chronic swelling and bleeding from the umbilicus, recurrent periorbital swelling and chronic infection of an ear tag wound. The lamb had a prolonged PT and APTT, and undetectable fibrinogen by clotting assay. A small amount of fibrinogen was measured on heat precipitation supporting a diagnosis of dysfibrinogenemia but this was not confirmed by measurement of fibrinogen antigen (due to lack of species-specific reagents).
Inhibitors
These are quite rare (or rarely documented/recognized) in animals. A suspected congenital or inherited defect in protein C has been reported in a Thoroughbred colt with recurrent thrombosis (Edens et al 1993). We have seen very low AT activity in a cat, which we suspect is congenital or inherited.
