Hematocrit (HCT) and packed cell volume (PCV) are used to measure red blood cell mass. An increase in red blood cell mass is equivalent to erythrocytosis and a decrease indicates an anemia. Because HCT and PCV are affected by changes in water, the HCT and PCV may be high due to water losses in a dehydrated animal (depending on the extent of dehydration). This can normalize a HCT/PCV in an anemic animal or may increase the HCT/PCV above the upper reference limit in a non-anemic animal, resulting in a relative erythrocytosis.
Method of measurement
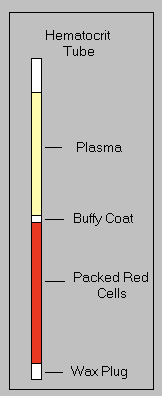
Although used synonymously, HCT and PCV actually represent different ways of measuring the proportion of blood composed of red blood cells.
- Hematocrit: This is actually a calculated value obtained from modern automated hematology analyzers. It is the product of the mean cell volume (MCV) and the red blood cell (RBC) count, both of which are directly measured by the analyzer. Therefore, if there are any inaccuracies in measurement of the MCV or RBC count, the HCT will reflect those inaccuracies. The formula used to calculate the HCT is as follows:
HCT = (MCV x RBC count)÷10
Thus, anything that falsely increases or decreases the MCV (e.g. storage of RBC may result in RBC swelling with an increased MCV, thus falsely increasing the HCT) or RBC count (e.g. hemolysis will decrease) will affect the HCT, but not necessarily, the PCV. - Packed cell volume (PCV): This is a directly measured value obtained from centrifuging blood in a microhematocrit tube in a microhematocrit centrifuge. The PCV is measured as the height of the red cell column in a microhematocrit tube after centrifugation (see image at right). It is the quickest and most readily available measure of the red blood cell component of blood. Unlike the HCT, this measurement is affected by plasma trapping and how the red blood cells pack within the column. At Cornell University, we centrifuge microhematocrit tubes for 10 minutes in ruminants versus 3 minutes in other species, because it is thought that red blood cells of ruminant do not “pack” as well as other species, however this remains to be tested. Examination of the “crit tube” can also provide subjective information about the color and clarity of the plasma (icterus, hemolysis, lipemia), and the size of the “buffy coat” (which contains WBC and platelets). Additionally, one can score and break the tube as desired to remove the plasma for refractometric protein estimation, or to extrude the buffy coat for smear-making. The “buffy coat smear” has the advantage of providing a concentrated preparation of nucleated cells, which can be useful if looking for low-incidence infectious agents (e.g., Anaplasma organisms in leukocytes).
Units of measurement
Both are expressed as % of the blood (SI units are L/L). The conversion formula to SI units is as follows
% ÷ 100 = L/L
Sample considerations
Sample type
Whole blood, body cavity fluids (PCV only)
Anticoagulant
EDTA is the preferred anticoagulant. Although citrate can be used, the volume of citrate in the tube (10% of the collection volume) will dilute the PCV or HCT accordingly. Heparinized whole blood can also be used.
Stability
HCT and PCV are optimally stable for 24 hours at 4°C. After this time, RBC tend to swell which increases the MCV, falsely increasing the HCT and PCV potentially (RBC do not pack as well). Also red blood cells start to hemolyze with storage, resulting in false decreases in both the HCT and PCV.
Site of collection
Studies in lactating and non-lactating dairy cows show an average difference in PCV of 4% and 2%, respectively, in blood taken from the jugular vein and mammary gland, with lower PCV in the mammary gland (Fisher 1962).
Interferences
- Lipemia, icterus: No effect.
- Hemolysis: Will decrease the HCT and PCV. This is considered an artifactual decrease with in vitro hemolysis. In an animal with true intravascular hemolysis versus in vitro hemolysis, the PCV or HCT is a better indicator of the oxygen carrying capacity of blood than the hemoglobin (which includes free hemoglobin and that within intact red blood cells, with the latter being the oxygen carriers).
Test interpretation

Increased values (erythrocythemia, erythrocytosis)
- Artifact: Insufficient centrifugation speed may falsely increase the PCV if the RBC do not pack properly. Studies in cows show a small difference in PCV with centrifugation times of 2 versus 4 minutes (decrease of 0-2% in 4 samples), with little change thereafter (Fisher 1962). With false increases in MCV (e.g. storage of blood), the HCT will be falsely increased, but usually do not exceed the upper reference limit (although this may mean a mildly anemic animal may no longer appear anemic). We have seen increases in MCV
- Physiologic: Some breeds of dogs may have higher RBC counts, hematocrit and hemoglobin concentration, such as Dachshunds (52% versus 48% HCT [Torres et al 2014]), Greyhounds (average 58-61% in 9-13 month old dogs [Shief et al 2007], average 59% in adult dogs [Campora et al 2011]) and Whippets (Uhriková et al 2014).
- Pathophysiologic (for more, refer to the erythrocytosis page)
- Relative change to blood water: Dehydration, splenic contraction secondary to epinephrine (e.g. horses).
- Absolute increase in RBC mass: Stimulated by erythropoietin (secondary erythrocytosis) or erythropoietin-independent (primary erythrocytosis or polycythemia vera).
Decreased values
- Artifact: Hemolysis of RBC due to sample collection or storage (in vitro hemolysis). In this setting, the measured hemoglobin is the most accurate measure of the animal’s oxygen carrying capacity and a HCT can be estimated by multiplying the hemoglobin x 3 (RBC contain 1/3 hemoglobin in most species). With false decreases in MCV (e.g. excess EDTA), the HCT will be falsely decreased, but usually the results are not below the lower reference limit.
- Pathophysiologic:
- Relative change to blood water: Over-dilution with fluids, splenic relaxation (anesthetic agents, tranquilizers).
- Absolute decrease in RBC mass: Indicates a true anemia, due to hemorrhage, hemolysis (intravascular, extravascular) or decreased production. Multiple mechanisms may be operative.
