Bicarbonate is the major extracellular buffer in the body. It is present in all body fluids and can be generated from CO2 and H2O in the presence of carbonic anhydrase. Bicarbonate on the chemistry panel gives an indication of acid-base status, but does not replace blood gas measurement as it does not supply information about the respiratory component of acid-base status or the pH of the animal. Bicarbonate values on a chemistry panel should always be interpreted with the anion gap, which is a calculated result, and the corrected chloride. The anion gap and assessment of strong ions (sodium and chloride, primarily) provides useful information for delineating causes of metabolic acidosis (loss or titration of bicarbonate) and can give you an indication of a mixed acid-base disturbance. Note, that this page refers to bicarbonate measurement with the chemistry analyzer and not a blood gas analyzer. For the latter, refer to laboratory detection page.
Method of measurement
The following method is used at Cornell University to measure bicarbonate on our automated chemistry analyzer.
Bicarbonate can also be measured on the blood gas analyzer, which uses ion selective electrodes to measure pH and hydrogen (and then calculates the bicarbonate from pH and hydrogen concentration).
Reaction type
Blanked end-point reaction
Procedure
This is a two step reaction that starts with phosphoenolpyruvate carboxylase (PEPC) catalyzing the oxidation of phosphoenolpyruvate (PEP) to oxaloacetate, in the presence of HCO3–. This first reaction is coupled to a second reaction that involves the transfer of a H+ from an nicotinamide adenine dinucleotide (NADH) analog to oxaloacetate using malate dehydrogenase (MDH); the coupling makes it a two point kinetic reaction (second order kinetics). The decreased concentration of NADH analog reduces absorbance at 415 nm, which directly correlates to the concentration of HCO3– in the sample.
The reactions are illustrated below:
PEP + HCO3 PEPC >→ Oxaloacetate + H2PO3
Oxaloacetate + NADH analog + H+ MDH > Malate + NAD+ analog
Units of measurement
The concentration of bicarbonate is is expressed in mEq/L (conventional units) and mmol/L (SI units). The conversion formula is essentially one to one or as shown below:
mEq/L x 1 = mmol/L
Sample considerations
Sample type for measurement on a chemistry panel
Serum and plasma.
Sample type for measurement on a blood gas panel
Heparin-anticoagulated blood (see laboratory detection page)
Anticoagulant
Heparin (lithium is usually used, but sodium or ammonium heparin can also be used)
Stability
Bicarbonate levels in human samples are stable for 7 days at 4 – 8°C and 40 hours at 15-25°C, when separated from erythrocytes and stored tightly sealed (per product sheet). Bicarbonate levels in serum stored at -20°C or -80°C are apparently stable for up to 6 months.
Interferences
- Lipemia: Severe lipemia (≥1500 lipemia index) may falsely decrease bicarbonate concentrations.
- Hemolysis: Marked hemolysis may decrease bicarbonate concentrations (≥600 hemolysis index)
- Icterus: Severe icterus may decrease bicarbonate concentrations (icteric index: >60 for conjugated and unconjugated bilirubin)
Test interpretation
Increased bicarbonate concentration

Increased levels of bicarbonate usually indicates a metabolic alkalosis and is usually secondary to chloride depletion, i.e. will be associated with a decreased strong ion difference (chloride disproportionately lower than sodium).
- False increase: Falsely increased HCO3− is quite rare but we occasionally see this in mailed-in samples from large animals (ruminants, particularly). We have also seen falsely increased bicarbonate with poor water quality supply to our chemistry analyzers. The reasons for this artifact are unknown. Very high concentrations of pyruvate and lactate dehydrogenase (LDH) secondary to severe muscle injury may falsely increase bicarbonate concentrations (Collins et al 1998) and decrease the anion gap, but this scenario is rare. The interference occurs because oxamate is added to the reaction to inhibit LDH (although not specified in the product information sheet), which can convert pyruvate and NADH to lactate and NAD. With very high concentrations of LDH, NADH is consumed, mimicking the second part of the reaction where MDH creates malate and falsely increasing bicarbonate concentrations, where are inversely proportional to the consumed NADH. In the three cases in the aforementioned report, LDH activity ranged from around 25,000-63,000/uL. We rarely ever see LDH activity >10,000 U/L (minimal increases in bicarbonate are seen at this LDH activity with or with added pyruvate) (Collins et al 1998).
- Iatrogenic: Administration of bicarbonate-containing solutions.
- Pathophysiologic: This usually indicates a metabolic alkalosis. This can be primary condition or secondary to a primary respiratory acidosis.
- Primary chloride-depleted metabolic alkalosis: This condition develops when HCl-rich gastric or abomasal secretions are lost in vomiting or are sequestered. This is the most common metabolic acid-base disturbance in adult dairy cattle and is infrequent in horses. It is commonly seen in dogs and cats with causes of vomiting (of gastric contents) and is often concurrently observed with a primary titration high anion gap acidosis from L-lactate production due to concurrent hypovolemia or the cause of the vomiting (e.g. uremic acidosis), i.e. there is a mixed disturbance. With a primary metabolic alkalosis, both H+ and Cl− are lost (loss of an acid = alkalosis). Gastric cells continue to produce HCl, a reaction that generates sodium bicarbonate in plasma (for more on the exact mechanism, refer to types of acid-base disturbances), resulting in a metabolic alkalosis. The net effect of acid (H+) loss and base gain (HCO3–) is an increase in blood pH, which may result in alkalemia (if pH increases above the reference interval for that species). The hallmarks of an uncomplicated metabolic alkalosis are an increased strong ion difference ([Na+ + K+]-Cl–), chloride is disproportionately low versus sodium and increased bicarbonate concentration (with the changes in bicarbonate being roughly proportional to the change in chloride). However, if there is a complicating concurrent metabolic acidosis (which frequently happens with disorders causing gastrointestinal HCl loss because of concurrent fluid losses leading to hypovolemia), then bicarbonate concentration may be normal. So the best laboratory indicator of a metabolic alkalosis is high strong ion difference or chloride being disproportionately low to sodium. In a metabolic alkalosis, the kidney should excrete the excess bicarbonate resulting in a high urine pH. However due to the hypochloremia, there is increased sodium delivery to the collecting tubules, the net effect of which is acid excretion (with bicarbonate retention). Both reduced NaCl (reduced salt sensing by macula densa) and hypovolemia (from fluid losses associated with the cause of vomiting or sequestration) will also stimulate aldosterone secretion, which will further promote acid loss from the kidney. Hypokalemia exacerbates these effects (see image above). The renal response to a chloride-depleted metabolic alkalosis is actually to excrete acid, resulting in a so-called paradoxic aciduria, because this is the last thing the kidney should be doing. So to help the kidney do its job to excrete excess bicarbonate in a chloride-depleted alkalosis, give the animal sodium chloride and supplemental potassium (since the latter is also low due to the primary disease). We see a paradoxic aciduria most commonly in cattle with primary metabolic alkalosis from dilated abomasa but it also has been reported in dogs.
- Causes of a primary chloride-depleted metabolic alkalosis are the following:
- Gastrointestinal losses or “sequestration” of chloride or hydrochloric acid:
- Vomiting: In small animals, vomiting due to pyloric obstruction, e.g. foreign body or some cases of gastric dilatation-volvulus, causes metabolic alkalosis. In contrast to gastric secretions, duodenal and pancreatic secretions are very rich in HCO3–. Therefore, if small intestinal contents as well as gastric secretions are lost in vomiting (which is more often the case), then metabolic acidosis, rather than alkalosis, develops.
- Gastric reflux in horses: This would lose hydrochloric-rich gastric secretions and is due to various causes. Ileus can also induce the same abnormalities. Metabolic alkalosis is uncommon in horses.
- Gastric obstruction, torsion or rupture: This can occur in dogs (e.g. gastric dilatation volvulus) and horses (e.g. secondary to proximal duodenal obstruction, enteritis or bloat).
- Abomasal atony, torsion or displacement, or “reflux”: Metabolic alkalosis is the most frequent acid-base abnormality in adult cattle and is usually due to either a displaced abomasum or abomasal atony (which can occur with vagus indigestion or in sick cattle with other disorders that do not primarily affect the gastrointestinal tract via unknown mechanisms). Any other disorder, in which abomasal outflow is impaired or obstructed. e.g. abomasal adhesions, abomasal inflammation, pressure on the abomasum from a fetus in advanced pregnancy or proximal duodenal obstruction (causes a severe metabolic alkalosis due to continued secretion of hydrochloric acid), can result in sequestration of HCl and metabolic alkalosis.
- Sweat loss: In horses, hypochloremia, hypokalemia, and metabolic alkalosis can develop when overexerted, through loss of water and chloride-rich electrolytes (KCl) in sweating. Increased distal delivery of sodium promotes acid excretion in the collecting tubules of the kidney.
- Renal loss: Loop diuretics can result in loss of excess chloride in relation to sodium (via inhibiting the Na-K-2Cl carrier, i.e. two chlorides are lost for one sodium) and a metabolic alkalosis may result because of increased sodium delivery to the collecting tubules (with resultant excretion of acid and retention of bicarbonate). This may also occur with thiazide diuretics, which block the NaCl carrier in the early distal nephron.
- Gastrointestinal losses or “sequestration” of chloride or hydrochloric acid:
- Causes of a primary chloride-depleted metabolic alkalosis are the following:
- Primary non-chloride-dependent metabolic alkalosis: This is a rare condition caused by excess aldosterone (which stimulates acid excretion in the collecting tubules). The alkalosis is generally mild unless there is concurrent hypochloremia.
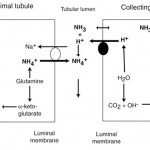
- Secondary or compensatory metabolic alkalosis: This is in compensation for a primary respiratory acidosis. In this case, the kidney excretes ammonium chloride (by generating ammonia from glutamine in the proximal tubules which combines with filtered chloride, which is then excreted, and by increasing hydrogen excretion in the distal tubules, see image below). New bicarbonate is generated in blood, resulting in a secondary or compensatory metabolic alkalosis. This offsets the acidemia that results from increased carbon dioxide from a primary respiratory acidosis. The kidney is so good at excreting ammonium chloride and promoting distal tubular acid excretion that, in the dog at any rate, it can actually correct the pH back to normal (given sufficient time, around 30 days) in a chronic respiratory acidosis (usually compensation does not return pH to normal).
- Primary chloride-depleted metabolic alkalosis: This condition develops when HCl-rich gastric or abomasal secretions are lost in vomiting or are sequestered. This is the most common metabolic acid-base disturbance in adult dairy cattle and is infrequent in horses. It is commonly seen in dogs and cats with causes of vomiting (of gastric contents) and is often concurrently observed with a primary titration high anion gap acidosis from L-lactate production due to concurrent hypovolemia or the cause of the vomiting (e.g. uremic acidosis), i.e. there is a mixed disturbance. With a primary metabolic alkalosis, both H+ and Cl− are lost (loss of an acid = alkalosis). Gastric cells continue to produce HCl, a reaction that generates sodium bicarbonate in plasma (for more on the exact mechanism, refer to types of acid-base disturbances), resulting in a metabolic alkalosis. The net effect of acid (H+) loss and base gain (HCO3–) is an increase in blood pH, which may result in alkalemia (if pH increases above the reference interval for that species). The hallmarks of an uncomplicated metabolic alkalosis are an increased strong ion difference ([Na+ + K+]-Cl–), chloride is disproportionately low versus sodium and increased bicarbonate concentration (with the changes in bicarbonate being roughly proportional to the change in chloride). However, if there is a complicating concurrent metabolic acidosis (which frequently happens with disorders causing gastrointestinal HCl loss because of concurrent fluid losses leading to hypovolemia), then bicarbonate concentration may be normal. So the best laboratory indicator of a metabolic alkalosis is high strong ion difference or chloride being disproportionately low to sodium. In a metabolic alkalosis, the kidney should excrete the excess bicarbonate resulting in a high urine pH. However due to the hypochloremia, there is increased sodium delivery to the collecting tubules, the net effect of which is acid excretion (with bicarbonate retention). Both reduced NaCl (reduced salt sensing by macula densa) and hypovolemia (from fluid losses associated with the cause of vomiting or sequestration) will also stimulate aldosterone secretion, which will further promote acid loss from the kidney. Hypokalemia exacerbates these effects (see image above). The renal response to a chloride-depleted metabolic alkalosis is actually to excrete acid, resulting in a so-called paradoxic aciduria, because this is the last thing the kidney should be doing. So to help the kidney do its job to excrete excess bicarbonate in a chloride-depleted alkalosis, give the animal sodium chloride and supplemental potassium (since the latter is also low due to the primary disease). We see a paradoxic aciduria most commonly in cattle with primary metabolic alkalosis from dilated abomasa but it also has been reported in dogs.
Decreased bicarbonate concentration
Unless there is clear evidence of sample collection or storage issues or a respiratory disorder that would cause accumulation of CO2, low bicarbonate is presumptive evidence of metabolic acidosis. Diseases that cause loss or titration of HCO3– must then be considered. Evaluation of the anion gap and corrected chloride concentration helps distinguish between these causes of a metabolic acidosis, although both may be occurring concurrently resulting in a mixed acid-base disorder. Metabolic acidosis is the most common acid-base disorder encountered in veterinary practice.
- False decrease: False decreases in bicarbonate occurs in aged samples. This is usually due to lactate production by cells with anaerobic metabolism, with buffering of bicarbonate in the plasma within the tube. Over-dilution with heparin and prolonged venous stasis will also decrease bicarbonate.
- Iatrogenic: Administration of ammonium chloride will cause a normal anion gap or strong ion (hyperchloremic) metabolic acidosis.
- Pathophysiologic: A decreased bicarbonate indicates a metabolic acidosis. This can be a primary disorder from loss or consumption of bicarbonate or gain of a chloride-containing acid or a secondary disorder in compensation for a primary respiratory alkalosis. These disorders can be distinguished by measuring the pH and partial pressure of carbon dioxide on a blood gas analysis, evaluating the animal for a respiratory disorder or cause of metabolic acidosis (see below) and examining the anion gap, strong ion difference, and evaluating changes in chloride with respect to sodium.
- Titration or consumption of bicarbonate metabolic acidosis: This is also called a high anion gap metabolic acidosis and is almost always a primary condition (it is never compensatory in response to a primary respiratory alkalosis). Bicarbonate consumption by organic acids that lack chloride as the anion (unmeasured anion) will increase the anion gap (and strong ion difference will be unchanged unless there is another acid-base abnormality or disease condition affecting strong ions). A titration or high anion gap acidosis is by far the most common cause of metabolic acidosis in small animals, camelids, horses and adult ruminants (rare in the latter). Acids that are unmeasured anions include lactate, noncarbonic acids (sulfates, phosphates and citrates), ketones (diabetic ketoacidosis), toxins (ethylene glycol and its metabolites, methanol) and drugs (acetylsalicylic acid). In these conditions, bicarbonate is consumed in buffering the accumulated noncarbonic acid, leaving the anion of the acid in its place (thus increasing the anion gap).
- Lactic acidosis: Usually results from decreased tissue perfusion with production of L-lactate (anaerobic glycolysis increases lactate production), e.g. hypovolemia, hypoxia. Lactic acidosis also results from grain overload in cattle. D-lactic acidosis is seen in small animals with diabetes mellitus and more frequently in calves.
- Acute kidney injury or chronic renal disease (usually the later stages) resulting in failure may result in a high anion gap metabolic acidosis due to retained noncarbonic acids that are normally filtered by the kidney (e.g. phosphates, citrates, hippurates).
- Ketoacidosis: Ketones (acetoacetic acid, acetone, β-hydroxybutyrate) accumulate in unregulated diabetic patients or camelids or ruminants with negative energy balance (e.g. transition dairy cows, camelids that are stressed, particularly in late pregnancy). The lack of insulin or insulin resistance in diabetes mellitus type I or II, respectively, leads to increased lipolysis of adipose tissue with enhanced presentation of non-esterified fatty acids (NEFA) to the liver. These fatty acids are used for energy, incorporated into lipoproteins (VLDL) or shunted into ketone production. In ruminants and camelids, negative energy balance causes excessive lipolysis, which is compounded by stress (hormones such as corticosteroids and epinephrine stimulate hormone-sensitive lipase which breaks down fat stores). Horses do not usually develop ketosis due to poorly developed ketone pathways.
- Toxicity: Dogs and cats with antifreeze (ethylene glycol) poisoning have a high anion gap metabolic acidosis caused by metabolites of ethylene glycol. They also have a high osmolal gap (measured minus calculated osmolality > 30) because ethylene glycol and its metabolites are unmeasured osmols but are not included in the equation to calculate osmolality (based on sodium, potassium, urea nitrogen and glucose).
- Strong ion metabolic acidosis: This is also called a normal anion gap or hyperchloremic metabolic acidosis because it is associated with a normal anion gap, low strong ion difference and chloride being disproportionately higher than sodium. This can be a primary or secondary disorder.
- Primary metabolic acidosis due to bicarbonate loss: Bicarbonate can be lost through the gastrointestinal tract or kidneys.
- Gastrointestinal tract: Vomiting of bicarbonate-rich solutions (biliary or pancreatic fluids), sequestration of bicarbonate within the gastrointestinal tract (e.g. ileus in horses), diarrhea (usually secretory), loss of saliva particularly in cattle (and to a lesser extent horses). Saliva is high in bicarbonate, so loss of saliva through inability to swallow (e.g. rabies, choke) can result in a metabolic acidosis. In these conditions, water and sodium are lost concurrently, hence hypovolemia ensues. As a response to hypovolemia, the kidney absorbs sodium and water. Sodium is absorbed with chloride, which then increases in blood, resulting in hyperchloremia with a normal anion gap.
- Kidneys: Filtered bicarbonate is usually absorbed (reclaimed) in the proximal renal tubules with filtered sodium or acids (e.g. phosphate, called titratable acidity) or ammonia generated by the proximal tubules or passively excreted by the distal tubules (see image above). Defective proximal renal tubule absorption of bicarbonate can cause a hyperchloremic bicarbonate loss acidosis, e.g. proximal renal tubular acidosis (also called Fanconi’s syndrome). Addison’s disease can also result in a hyperchloremic metabolic acidosis because aldosterone promotes hydrogen excretion in the distal tubules of the kidneys (lack of aldosterone would promote hydrogen retention or an acidosis). Distal renal tubular acidosis will also result in a normal anion gap hyperchloremic metabolic acidosis due to defective excretion of hydrogen by distal tubules.
- Primary metabolic acidosis due to loss of high SID fluids: Fluids which contain more sodium than chloride (potentially secretory diarrhea) can result in a normal anion gap acidosis, with a decreased SID in plasma or serum.
- Primary metabolic acidosis due to gain of a chloride-containing acid: This occurs in disorders where there is defective renal excretion of hydrogen in the distal part of the kidney, e.g. distal renal tubular acidosis.
- Secondary strong ion metabolic acidosis: This occurs when the kidney reduces acid excretion (by decreasing ammonia generation in the proximal convoluted tubules or H+ATPase activity in the collecting tubules, subsequently causing filtered bicarbonate to be excreted and chloride to be retained) because of a primary respiratory alkalosis. The kidney also promotes bicarbonate loss by stimulating the pendrin pump (chloride/bicarbonate exchanger) in the apical membrane of type B intercalated cells. This excretes bicarbonate in the urine in exchange for chloride. The kidney is so effective at excreting bicarbonate when compensating for a primary respiratory alkalosis, that this compensatory response can actually correct the pH (given sufficient time, around 14 days) in dogs. It is unknown if this happens in other species. For more information on how the kidney handles bicarbonate absorption and hydrogen excretion, refer to the renal portion of this website.
- Primary metabolic acidosis due to bicarbonate loss: Bicarbonate can be lost through the gastrointestinal tract or kidneys.
- Titration or consumption of bicarbonate metabolic acidosis: This is also called a high anion gap metabolic acidosis and is almost always a primary condition (it is never compensatory in response to a primary respiratory alkalosis). Bicarbonate consumption by organic acids that lack chloride as the anion (unmeasured anion) will increase the anion gap (and strong ion difference will be unchanged unless there is another acid-base abnormality or disease condition affecting strong ions). A titration or high anion gap acidosis is by far the most common cause of metabolic acidosis in small animals, camelids, horses and adult ruminants (rare in the latter). Acids that are unmeasured anions include lactate, noncarbonic acids (sulfates, phosphates and citrates), ketones (diabetic ketoacidosis), toxins (ethylene glycol and its metabolites, methanol) and drugs (acetylsalicylic acid). In these conditions, bicarbonate is consumed in buffering the accumulated noncarbonic acid, leaving the anion of the acid in its place (thus increasing the anion gap).
