Automated hematologic analyzers (electrical impedance- or laser-based) can measure platelet volume (MPV), just like they can measure red blood cell volume (MCV). The MPV can be useful for distinguishing between causes of thrombocytopenia (just like the MCV can provide guidance as to causes of anemia), but should never be used in isolation. During active thrombopoiesis, larger platelets than normal may be produced (similar to immature anucleate RBC, which are usually larger than mature RBC). These larger platelets can be observed in blood smears and, if in sufficient number, will increase the MPV above reference intervals. Large platelets are generally presumed to be young platelets, however, large size is not synonymous with immaturity. Also, there is a substantial degree of variation in platelet size in healthy animals, particularly in some species, such as cats, so identification of large platelets in a thrombocytopenic animal depends on the species, number and appearance of the platelets.
Method of measurement
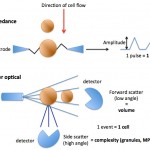
This can be done with impedance or optical based analyzers:
- Impedance: The platelet volume is determined by the change in resistance as platelets pass through an apeture containing an electric field in single file. The degree to which the platelets alter the resistance as they pass through the field corresponds to their size (specifically volume).
- Laser: The platelet volume is measured by the degree to which the cells scatter light in a forward direction (low angle light scatter). Optical analyzers can also provide a measure of the coefficient of variation (mean/standard deviation) of the platelet volume or platelet distribution width (analogous to the red blood cell distribution width or RDW), although this value is seldom reported (or even used in the laboratory).
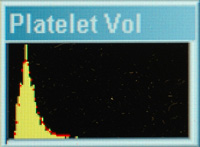
With both techniques, the different sized platelets in the sample can be represented on a frequency distribution curve of proportion of platelets versus platelet volume and the analyzer can provide results for mean volume of platelets (MPV). The histogram curve is usually asymmetrical (most platelets are small versus large in size).
Units of measurement
Femtoliters (fL)
Test interpretation
A low MPV is of no diagnostic relevance. The MPV should not be interpreted alone but in conjunction with the platelet count. An increased MPV, with the presence of large platelets in a blood smear and absence of platelet clumps, in a thrombocytopenic animal is suggestive of a bone marrow response to the thrombocytopenia (a regenerative thrombocytopenia); however the lack of large platelets does not exclude a bone marrow response and large platelets can be present due to abnormal bone marrow production of platelets. Causes of an increased MPV are given below. Also note, that low numbers of large platelets may not increase the MPV (which represents the mean) above reference intervals.
Increased MPV
- Artifact: Platelets that are activated either degranulate or imbibe water. This causes them to look pale and agranular in a blood smear and is helpful for distinguishing between artifactual and other causes of increased platelet volume
- Storage of blood: Platelets activate and swell with storage, particularly at 4°C (the preferred temperature of storage of EDTA blood for a hemogram). Thus platelet counts and the MPV are most valid in freshly collected and analyzed samples.
- Platelet clumping: Smaller clumps that fall within the counting size threshold are “seen” as single large platelets by most automated analyzers. Thus clumps will decrease the platelet count and increase the MPV. This is why blood smears should be examined for platelet clumping to verify any platelet count.
- Physiologic: All animals to some extent have large platelets, but this is particularly true of cats, who have a much higher upper reference limit (and broader reference interval) for MPV than other species. The variability of platelet size in cats may be due to genetic variation in the structure of β1-tubulin (Boudreaux et al 2010). Platelets should have normal granularity (note, equine platelets have less obvious granules than other species).
- Pathophysiologic
- Regenerative thrombocytopenia: As indicated above, increased megakaryopoiesis with increased platelet production can result in the generation of platelets that are larger than normal (and contain more RNA than normal platelets).
- Defective platelet production: Abnormal platelet production due to defective megakaryocytes can result in a high MPV. Causes of abnormal platelet production are the following:
- Inherited defect in dogs: β1-tubulin defect in Cavalier King Charles spaniels (CKCS), Norfolk and Cairn Terriers and other breeds. Tubulin is required for fragmentation of the megakaryocyte cytoplasm (which is how platelets are produced). Abnormal tubulin will result in decreased fragmentation and lower numbers of larger platelets. These platelets are of normal function and affected dogs are asymptomatic despite low platelet counts.
- Hematopoietic neoplasia: Myelodysplastic syndrome, acute myeloid leukemia (particularly megakaryocytic leukemia). Due to the genetic mutation and neoplastic transformation, abnormal platelets are produced. Unlike inherited defects, platelets are discernibly abnormal in blood smears (i.e. they are dysplastic).
Related links
- See Platelet number under Hemostasis.
- Further discussion on thrombocytopenia and thrombocytosis is found under disorders of platelet number.