Interpretation
Neoplasia with endocrine/neuroendocrine appearance; metastatic medullary (C-cell) thyroid carcinoma suspected
Explanation
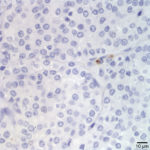
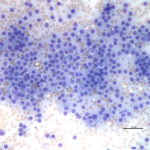
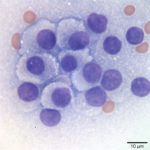
The aspirate fits with an endocrine/ neuroendocrine, “naked nuclei” cytomorphology (Question 1). Characteristically, aspirates are highly cellular, with numerous round, bare nuclei scattered over a background of disrupted cytoplasm. Nuclei typically retain a crisp outline, and are not smudged. When intact, cells are round to polygonal and have variably distinct cytoplasmic borders, and may be found in groupings of loosely adherent cells. Anisocytosis and anisokaryosis are often minimal to mild, but not necessarily predictive of benign biologic behavior. However, marked atypia indicates a carcinoma. These neoplasms typically originate from endocrine or neuroendocrine tissue, and examples include thyroid and parathyroid neoplasms, pheochromocytomas, paragangliomas (including chemodectomas such as aortic body tumors or carotid body tumors), islet cell tumors, and carcinoids. Anal sac adenocarcinomas of dogs often manifest this cytologic pattern when aspirated.
The ventral and paratracheal location made a paraganglioma less likely (Question 2). These are most commonly found at the base of the heart or close to the mandible at the carotid bifurcation (chemodectomas). Thus, a thyroid or parathyroid tumor was most probable given the location. Parathyroid tumors are typically small, benign, and functional – their exuberant excretion of parathyroid hormone leads to hypercalcemia, which may be found incidentally on a chemistry panel, or may cause clinical signs such as polyuria and polydipsia. Therefore, the large size, presumed metastasis, and unremarkable calcium profile made a parathyroid tumor less likely (Question 3). Thyroid tumors may arise from follicular or medullary (parafollicular, C-cell) cells, both of which may exhibit an endocrine/neuroendocrine appearance. Either may contain pink extracellular material consistent with colloid – produced constitutively by the follicular tumor or aspirated from adjacent or entrapped non-neoplastic follicles in either tumor. Follicular thyroid cells may contain blue-black cytoplasmic pigmentary granules (presumed tyrosine granules), which are not expected with medullary tumors. Neither colloid nor pigmentary granules were appreciated in the lymph node aspirate. Although there is considerable morphologic overlap between follicular and medullary thyroid cells and they may appear nearly identical, the relatively discohesive and distributed intact cells, and lack of blue/black pigment suggested a medullary C-cell origin (Figs. 1-2).
 |
 |
Additional Information
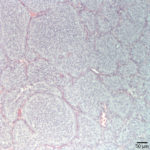
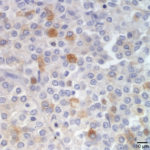
Both the tumor and the affected lymph node were excised and submitted for histopathology. The thyroid was effaced by a densely cellular, poorly demarcated and infiltrative neoplasm made up of round to polygonal cells forming sheets, lobules and pseudorosettes. The tumor demonstrated capsular, lymphatic, and vascular invasion, with surgical margins of <0.5 mm. The lymph node was effaced by a similar population of neoplastic cells (Figure 3). Approximately 30% of the tumor cells within the primary thyroid mass demonstrated positive cytoplasmic immunoreactivity for calcitonin (a medullary cell marker – Figure 4), but were diffusely negative for thyroglobulin (a follicular cell marker – Figure 5).
Diagnosis: Medullary (C-cell) thyroid carcinoma with metastasis to the retropharyngeal lymph node
Discussion
Most thyroid carcinomas may be sorted into two large categories – those of follicular cell origin or medullary (C-cell) origin.1 Medullary thyroid carcinomas (MTC) are clinically found most commonly in dogs, bulls, and horses with isolated reports from non-domestic species such as the red fox,2 hedgehog,3 mouflon,4 and ferret.5 MTC may follow C-cell hyperplasia (often secondary to high dietary calcium intake in bulls), occur sporadically, or arise as part of a hereditary syndrome. In people, hereditary MTC is often found in conjunction with pheochromocytoma and parathyroid adenoma or hyperplasia as part of a multiple endocrine neoplasia syndrome (MEN2). Nearly all patients have activating genetic mutations within the RET (Re-arranged during Transfection) proto-oncogene, which codes for a transmembrane receptor-type tyrosine kinase expressed on cells emanating from the neural crest, branchial arches, or urogenital system.6 Although familial MTC has been reported in bulls and dogs, the underlying genetic mutation has not been established.7
The signature secretory molecule of normal or neoplastic C-cells is the hormone calcitonin. Calcitonin depresses the action of osteoclasts to decrease serum calcium levels. It has a role in regulating bone turnover and calcium homeostasis, and its production increases in pregnancy and lactation.8 Paradoxically, knockout mice without the gene for calcitonin have enhanced bone mass (would have expected osteopenia due to excessive osteoclast bone resorption).9 Elevated levels of serum calcitonin are found with MTC, and reflect the tumor burden in people.6 Unfortunately, assays for measuring serum calcitonin are not readily available for dogs. Remarkably, even with high levels of calcitonin, only very rarely are serum calcium levels abnormal in either people or dogs with MTC. Older reports in dogs uncovered a few cases of concurrent hypocalcemia,10-11 but most dogs have unremarkable serum calcium levels when evaluated (as did our patient). Osteoclasts rapidly become refractory to calcitonin, likely by decreasing expression of the calcitonin receptor,12 which may stabilize serum calcium concentrations. Both human and canine patients retain normocalcemia after bilateral thyroidectomy, and do not require supplemental calcitonin.
Although serum calcitonin assays are not readily accessible, calcitonin expression probed by immunohistochemistry (IHC) on incisional or excisional biopsy specimens is routine. Thyroid carcinomas are considered MTC with positive calcitonin expression and negative thyroglobulin expression; follicular thyroid carcinomas (FTC) are calcitonin negative and thyroglobulin positive. Thyroid carcinomas that express neither13 or both14 of these markers have been reported in dogs, but these types of tumors are not well defined, even in human medicine. Other immunohistochemical markers such as neuron specific enolase (NSE) for neuroendocrine tumors like MTC or thyroid transcription factor-1 (TTF-1) for FTC, are less specific and may be expressed by either FTC or MTC to a varying degree.15 IHC characterization is important as the solid/ compact type of FTC appears very similar to MTC when stained with routine histochemical stains like hematoxylin and eosin (H&E). Compact FTC resembles neuroendocrine tumors with solid sheets of cells forming minimal follicles,1and MTC may harbor entrapped non-neoplastic resident follicles or form pseudofollicles, mimicking FTC.16 Based on H&E staining, MTC was once thought to be rare in dogs, comprising 2-3% of thyroid carcinomas.10 With application of calcitonin and thyrogobulin IHC, presumptive solid/compact FTC often gets reclassified to MTC, and in several studies, the incidence of MTC shifted from under 5% to over 20% of canine thyroid carcinomas.15,17
Accurate differentiation of follicular from medullary thyroid carcinoma is required to determine if the tumors diverge in underlying cause, prognosis, or treatment requirements. In general, thyroid carcinomas tend to metastasize to the regional lymph nodes and lung, with other distant metastasis less common. MTC have been reported to be less locally invasive and thus potentially more resectable than FTC,18 but this did not correspond to a significant difference in overall survival, disease-free survival, or time to metastasis after surgery.17 However, the surface expression of molecular targets such as vascular endothelial growth factor, cyclooxygenase-2, or P-glycoprotein is more prevalent in MTC, suggesting potential differences in response to chemotherapeutic agents.19
Follow-up
The patient received definitive radiation therapy targeting the resection sites, followed by chemotherapy with toceranib (PalladiaTM). The patient requires thyroxine supplementation for hypothyroidism, presumably induced by radiation therapy treatments. Approximately 8 months after surgery, she is reportedly doing very well at home, and imaging shows no evidence of tumor regrowth or metastasis.
References
- Rosal TJ and DJ Meuten. Tumors of the Endocrine Glands. 2017. In: Tumors of Domestic Animals. Meuten D. 5th ed, pp. 803-809. John Wiley & Sons, Inc.
- Hirayama, Y. Kagawa, K. Nihtani, H. Taniyama. Thyroid C-cell Carcinoma with Amyloid in a Red Fox (Vulpes vulpes schrenchki). Vet Pathol 1999; 36(4): 342 –344.
- Miller DL, Styer EL, Stobaeus JK, Norton TM. Thyroid C-cell carcinoma in an African pygmy hedgehog (Atelerix albiventris). J Zoo Wildl Med 2002; 33(4): 392–396.
- LA Griner. Thyroid medullary carcinoma and pheochromocytoma in an aged European mouflon (Ovis ammon musimon). J Zoo An Med 1981; 12: 117-120.
- Fox JG, Dangler CA, Snyder SB et al. C-Cell Carcinoma (Medullary Thyroid Carcinoma) Associated with Multiple Endocrine Neoplasms in a Ferret (Mustela putorius). Vet Pathol 2000; 37(3): 278 – 282.
- Wells, SA et al. Revised American Thyroid Association Guidelines for the Management of Medullary Thyroid Carcinoma: The American Thyroid Association Guidelines Task Force on Medullary Thyroid Carcinoma. Thyroid 2015; 25(6): 567-610.
- Lee JJ et al. A dog pedigree with familial medullary thyroid cancer. Int J Oncol 2006 29:1172-1182.
- Felsenfield AJ and Levine BS. Calcitonin, the forgotten hormone: does it deserve to be forgotten? Clin Kidney J 2015; 8: 180-187.
- Hoff AO et al. Increased bone mass is an unexpected phenotype associated with deletion of the calcitonin gen. J Clin Invest 2002; 110 (12): 1849-1857.
- Leav I et al. Adenomas and carcinomas of the canine and feline th Am J Pathol 1976; 83:61-93.
- Patnaik AK, Lieberman RA, Erlandson WM, Acevedo WM, and Liu SK. Canine medullary carcinoma of the thyroid. Vet Pathol 1978; 15: 590-599.
- Takahashi S et al. Downregulation of calcitonin receptor mRNA expression by calcitonin during human osteoclast-like cell differentiation. J Clin Invest 1995; 95(1):167-171.
- Soler Arias EA, Castillo VA, and Caneda Aristarain ME. Calcitonin-negative primary neuroendocrine tumor of the thyroid (nonmedullary) in a dog. Open Vet J 2016; 6(3): 223-227.
- Brown PJ, Rema A, and Gartner F. Thyroglobulin, Calcitonin, Ki 67 and CEA immunohistochemical staining of dog thyroid carcinomas. Eur J Vet Pathol 2001; 7, 11-16.
- Ramos-Vara JA, Miller MA, Johnson GC, et al. Immunohistochemical detection of thyroid transcription factor-1, thyroglobulin, and calcitonin in canine normal, hyperplastic, and neoplastic thyroid gland. Vet Pathol 2002; 39:480–487.
- Leblanc B, Parodi AL, Lagadic M, et al. Immunocytochemistry of canine thyroid tumors. Vet Pathol 1991; 28:370–380.
- Campos M, Ducatelle R, Kooistra HS et al. Clinical, pathologic, and immunohistochemical prognostic factors in dogs with thyroid carcinoma. J Vet Intern Med 2014;28:1805–1813.
- Carver JR, Kapatkin A and Patnaik AK. A comparison of medullary thyroid carcinoma and thyroid adenocarcinoma in dogs: A retrospective study of 38 cases. Vet Surg 1995; 24:315– 319.
- Campos M, Ducatelle R, Kooistra HS, et al. Immunohistochemical expression of potential therapeutic targets in canine thyroid carcinoma. J Vet Intern Med 2014;28(2):564-570.
Authored by: LS Kelly