Interpretation
Intracytoplasmic inclusions compatible with canine distemper virus (CDV) infection.
Explanation
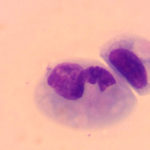
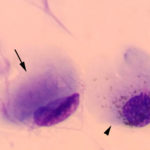
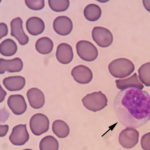
The conjunctival swabs contained low numbers of squamous epithelial cells, some of which contained melanin pigment (Figure 2a). There was no evidence of an inflammatory infiltrate. Several cells contain large grainy light pink intracytoplasmic inclusions (Figure 1-2a, Question 1). Similar inclusions were observed in low numbers of lymphocytes in a peripheral blood smear (Figure 3). These findings, with the associated clinical signs of ocular, neural and respiratory symptoms (non-responsive to antibiotics) led to a presumptive antemortem diagnosis of CDV infection. The cytoplasmic inclusions are paler than normal for CDV, which usually stain more magenta and are more prominent with rapid stains, like Diff-quik®, than Wright’s-stained smears. With the latter stain, the inclusions are often a light pale grey-blue and can be overlooked, particularly if they are only present in low numbers of cells (for representative pictures of Wright’s and Diff-quik-stained CDV inclusions in blood cells, view our infectious agent [blood] image gallery). The reason for the lighter staining of the inclusions in this case is unknown.
Discussion
Canine distemper virus is a highly contagious pathogen of dogs, causing infectious outbreaks of respiratory, ocular, and neurologic disease.1 Canine distemper virus is a Morbillivirus and part of the Paramyxoviridae family. The virus is mostly spread via aerosolization, being shed primarily in respiratory secretions, although transplacental infection does occur and other body fluids, such as urine and feces, also contain infectious virions.1,2 Infected animals that survive acute infection or that are infected by mild virulent strains develop life-long immunity, but shedding can be prolonged (up to 3 months).1 Crowding and close contact between animals, combined with the availability of a naïve and susceptible dog population (such as puppies in a pet shop), increase the risk of infection. Puppies between 2-3 months of age are particularly susceptible due to declining maternal immunity.1 Although the virus is primarily considered to be a canine pathogen, there are many different strains and it is now recognized that CDV is not host specific and frequently crosses species boundaries.3,4 In fact, the virus is somewhat of an “emerging” disease, with appearance of new virus strains of variable pathogenicity in dogs5 and appearance of specific strains in wildlife species, some of which are endangered, such as elephants and giant pandas. The virus also demonstrates variable pathogenicity with higher mortality in some species (e.g. mortality approaches 100% in ferrets, which are used as an experimental model of the virus) than others.2,3 Domesticated dogs are thought to be the primary reservoir of infection for wildlife, although the virus has been identified in various wildlife species and wildlife reservoirs of infection certainly exist.2,3
This puppy demonstrated some of the classic clinical signs of acute CDV infection, including fever and ocular (chemosis), neurologic (rhythmic jaw movements) and respiratory (bilateral nasal discharge and dyspnea) signs. Dogs with acute infection can also develop diarrhea with attending sequelae of dehydration and hypovolemia. Neurologic symptoms are variable, ranging from seizures to cerebellar dysfunction to paresis to myoclonus, and can occur with or after resolution of acute infection. Central nervous system signs are thought to be a consequence of viremia with secondary invasion into brain and spinal cord cells. Hyperkeratosis of foot pads and nasal cutaneous epithelium can be a feature of CDV infection and manifest along with neurologic signs, due to virus proliferation in the dermal epithelium.1
Hematologic changes with acute CDV infection are variable, however lymphopenia is a characteristic finding (Question 2).1,2,6,7 The lymphopenia has been attributed to lymphocytolysis, apoptosis and decreased proliferation and is associated with peak viremia and lymphoid depletion in tissues.2,6 The virus invades lymphocytes via the signaling lymphocyte activation molecule (CD150/SLAM).1 In experimental studies, both T (with no selectivity for helper or cytotoxic T cells) and B cells are reduced, however the depletion of B lymphocytes is more profound and longer-lasting (at least 24 days).6 Lymphoid depletion has been associated with immunosuppression, increasing susceptibility to secondary bacterial infections, particularly pneumonia. This puppy was borderline lymphopenic per provided reference intervals, however this case illustrates the limitations of reference intervals, which are usually established for adult dogs (>1 year of age). Puppies have higher lymphocyte counts than adult dogs at this age (mean ± 2 SD of 5.1 ± 3.0 thou/μL), which could be a consequence of an epinephrine response (physiologic lymphocytosis).7 Thus, this puppy was likely lymphopenic when taking its age into account. Anemia is also commonly found with CDV infection and may be secondary to inflammatory disease, although direct virus effects on erythroid production are possible, since inclusions are found in erythrocytes in blood and bone marrow.7 Hematocrit can also be lower in puppies of this age, but the hematocrit in this puppy was below reported values.7,8 Neutropenia has been documented in some cases of CDV, although this is not a consistent finding. Virus inclusions are found in neutrophils, which could reflect virus effects on production (this has not been documented), however concurrent consumption due to a secondary bacterial infection is possible and could explain the neutropenia in the puppy in this report. A mild to moderate thrombocytopenia has been reported secondary to CDV infection, usually during the acute viremic phase,9 or after vaccination with modified-live vaccines.10 The thrombocytopenia in experimental infection has been attributed to immune-mediated destruction and decreased production secondary to virus infection of marrow megakaryocytes.9 This puppy was not thrombocytopenic per the provided reference interval, however the lower limit of the provided interval is lower than our lower reference limit and the puppy could be thrombocytopenic. It is also possible that microclots formed during sample collection if venipuncture was difficult in this puppy. This pre-analytical artifact would falsely decrease all counts in blood.
Puppies of this age can be mildly microcytic, which may be secondary to a physiologic iron deficiency (low stores combined with rapid growth) (Question 3). However, the microcytosis is more severe than prior reported values for this age in other breeds.7 There was no evidence of gastrointestinal blood loss and the high MCHC does not support a true iron deficiency as a contributing cause of the microcytosis or the anemia. Microcytosis can also be a symptom of portosystemic shunts, which is thought to be due to a “functional” iron deficiency from abnormal iron metabolism (cause unclear). We have also observed lower MCVs in adult dogs of this breed, so the microcytosis could be a breed-related finding. Although there are other causes of microcytosis (see MCV under the hematology section), one cause that may definitely be occurring in the dog is a pre-analytical error of “short sampling” in this young puppy. A small volume of blood in a large EDTA tube (excess EDTA) causes shrinkage of erythrocytes, with a consequent reduction in MCV and increase in MCHC. The high normal MCHC in this puppy suggests that this may have been occurring. We frequently see this artifact in samples from cats or other species, particularly if the EDTA tube (even a microtainer) is <1/4 full.
Diagnosis of CDV is facilitated by characteristic clinical signs and detection of the characteristic inclusion bodies in peripheral blood cells (neutrophils, lymphocytes, monocytes, erythrocytes), cerebrospinal fluid, or infected epithelial tissues. Conjunctival mucosa is easy to sample and is the most frequently examined epithelial tissue for inclusions, although inclusions can also be seen in urinary bladder and cutaneous epithelium on histologic assessment of tissues.1 Inclusions occur within the nucleus and cytoplasm, but the cytoplasmic inclusions are easier to detect, particularly when using rapid stains. Other stains, including new methylene blue, have been used to detect inclusions, which consist of viral nucleocapsids.1,7 The sensitivity of cytologic detection of inclusions for the diagnosis of CDV is unknown. Inclusions can be readily missed (particularly if low in number or if there is a low clinical index of suspicion) and they need to be distinguished from other substances, e.g. ophthalmic ointment in conjunctival swabs, or staining variation. Immunofluorescent or immunohistochemical detection of CDV antigen is required to confirm that inclusions are due to CDV, but inclusions, particularly in histologic sections, does not mean that the clinical disease is due to CDV. They just mean prior infection. Other methods, including serologic testing for anti-virus antibody (e.g. serum neutralization, ELISA) and PCR can be used to diagnose infection (which does not always mean disease, since vaccine responses or strains can be similarly identified) in histologic tissues and body fluids (including feces). High serum neutralization titers do correlate to protection in vaccinated animals (albeit against known strains). Specific PCR primers or virus sequencing can be used to distinguish between vaccinal and wild type strains or identify the origin of a particular strain in an outbreak.1,5
Further information and follow up
The puppy was treated symptomatically with antibiotics, intravenous fluids, nebulization and oxygen therapy. There was no improvement over 48 hours and the puppy developed myoclonus. The puppy was subsequently humanely euthanized. Gross post-mortem examination revealed firm mottled lungs with coalescing tan nodules. Histologic examination of post-mortem tissues revealed moderate to severe necrotizing bronchopneumonia with mixed inflammatory infiltrates (neutrophils, lymphocytes, macrophages with no bacteria), lymphoid depletion in the spleen, thymus and multiple lymph nodes, and nuclear and cytoplasmic inclusions in lymphocytes, various epithelia (bile duct, renal tubules, trachea, urinary bladder) and brain (neurones and glial cells). The post-mortem findings were consistent with CDV infection.
References
- Green CE and Vandevelde M. 2012. Canine Distemper. In: Greene: Infectious Diseases of the Dog and Cat. 4th ed. Green CE, ed. Elsevier; St Louis, MO. pp 25-43.
- Zhao et al. 2015. Pathogenesis of canine distemper virus in experimentally infected raccoon dogs,
foxes, and minks. Antiviral Res 122:1-11. - Martinez-Guiterrez and Ruiz-Saenz. 2016. Diversity of susceptible hosts in canine distemper virus infection: a systemic review and data synthesis. BMC Vet Res 12:78.
- Viena et al. 2015. Dynamics of a morbillivirus at the domestic-wildlife interface: Canine distemper virus infection in domestic dogs and lions. Proc Natl Acad Sci U.S.A. 112:1464-9.
- Riley and Wilkes. Sequencing of emerging canine distemper strain reveals new distinct genetic lineage in the United States associated with disease in wildlife and domestic canine populations. Virol J 12:219.
- Schobesberger et al. 2005. Canine distemper virus-induced depletion of uninfected lymphocytes is associated with apoptosis. Vet Immunol Immunopathol 104: 33-44.
- Jain N. 1986. In: Schalm’s Veterinary Hematology. 4th ed. Lea & Febiger; Philadelphia, PA. pp 1046-8 and 1130-3 (Case 37) for distemper and pp 104-25 for age-associated hemogram changes.
- Harper et al. 2003. Age-related variations in hematologic and plasma biochemical test results in Beagles and Labrador Retrievers. J Am Vet Med Assoc 223:1436-42.
- Axthelm and Krakowka. 1987. Canine distemper virus-induced thrombocytopenia. Am J Vet Res 48:1269-75.
- Stokol and Parry. 1997. The effect of modified live virus vaccination on von Willebrand factor antigen concentrations and platelet counts in dogs. Vet Clin Pathol 26: 135-7.
Authored by: T Stokol.
Acknowledgements: Many thanks to Dr. C. Narbus for provision of photomicrographs, clinical case material and post-mortem findings. This case would not have been presented without you.