Interpretation
Pleural fluid: Chylous effusion
Pericardial fluid: Lymphoma, large cell
Explanation
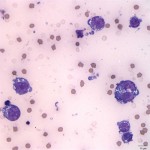
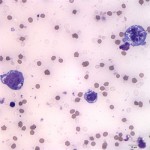
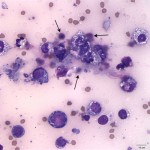
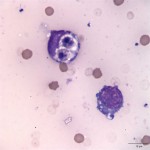
The pleural effusion consisted of 88% lymphocytes with a mixture of small and intermediate forms (Question 1) with fewer non-degenerate neutrophils, and rare macrophages (Figure 4). The predominance of small and intermediate lymphocytes is consistent with a chylous effusion (Question 2), despite the absence of the classic milky white appearance of the fluid. The medium orange color was likely due to the presence of the red blood cells. The pericardial fluid smears consisted of many large neoplastic lymphocytes, unlike the small and intermediate sized lymphocytes present in the pleural effusion (Question 3, Figure 5). This was consistent with a diagnosis of large cell lymphoma (Question 4). The neoplastic lymphocytes had a high nuclear to cytoplasmic ratio with slightly increased amounts of deep blue cytoplasm, which contained numerous small clear discrete cytoplasmic vacuoles. The cells had a round centrally to paracentrically located nucleus with finely stippled chromatin and 1-5 distinct nucleoli. They displayed moderate anisocytosis and mild anisokaryosis. There were frequent binucleate forms and low numbers of mitotic figures were noted (Figure 6). Necrotic cellular debris was present in the background as well (Figure 7). Cytophagic activity was also rarely noted (presumptive cannibalism, Figure 8). These neoplastic cells were not noted in the pleural fluid.
Further case information
A Cardiology consult was performed while the cat was in the hospital. An echocardiogram revealed an enlarged left atrium with a thickened left ventricular free wall, which was consistent with hypertrophic cardiomyopathy. The cat was started on a regimen of cardiac medications for heart failure including furosemide, benazepril, and baby aspirin. The Oncology service consult found the cat to be in poor body condition (3/9) with no abnormalities noted on abdominal palpation and no peripheral lymphadenopathy. They recommended full staging and treatment with the CHOP chemotherapy protocol (cyclophosphamide, doxorubicin, vincristine, prednisolone). All further diagnostics and treatment options were declined by the owner at that time. The cat was started on prednisolone for palliative care of the lymphoma.
Discussion
Unlike dogs, the finding of small amounts of pericardial effusion is a relatively common finding in cats with cardiac disease.1 It is also uncommon for cats to be symptomatic due to their pericardial effusion.2 In dogs, pericardial effusion is most commonly associated with neoplasia or idiopathic pericarditis and frequently results in cardiac tamponade with associated clinical signs.1,3 One study of feline necropsies showed that most cases of pericardial effusion in cats were part of a more generalized disease with congestive heart failure (CHF) and feline infectious peritonitis (FIP) being the most common diseases.2 In a more recent retrospective study of 146 cats with pericardial effusion, congestive heart failure was by far the most common cause of pericardial effusion (75%), whereas there was only one case with suspected FIP.1 The authors hypothesized the under-representation of FIP (and potentially neoplastic or other systemic disease) cases may have been due to the lack of necropsies in which a specific search for pericardial disease or effusions was performed. Of the cats with CHF in this study, hypertrophic cardiomyopathy (HCM) was the most common underlying disease.1 HCM is a disease of the ventricular (primarily left ventricular) myocardium characterized by mild to severe thickening of the papillary muscles and wall and is the most commonly diagnosed cardiac disease in cats.4 Along with pulmonary edema, pleural effusion is also common in cats with heart failure and can be a high protein transudate or a chylous effusion.4 In fact, the most common cause of chylothorax in cats is heart failure. Of the pleural effusion samples submitted for cytology in the previously mentioned retrospective study, chylous effusion, modified (high protein) transudate, and transudate were the most common cytologic diagnoses.1 Two cats had pericardial effusion samples evaluated, one of which was chylous and the other was exudative with mesothelial hyperplasia and dysplasia.1
Neoplasia of the heart and pericardium is rare in dogs and even more rare in cats.5 Primary cardiac tumors are rare in the cat, but the following have been reported: mesothelioma, primary cardiac hemangiosarcoma, aortic body tumors, myxoma, and rhabdomyosarcoma.5 Metastatic tumors to the heart are more common and include lymphoma, mammary gland carcinoma, pulmonary carcinoma, salivary gland adenocarcinoma, oral melanoma, rhabdomyosarcoma, sweat gland adenocarcinoma, squamous cell carcinoma, and mast cell tumor.5 Overall, the most common primary or metastatic cardiac tumor in cats is lymphoma and, in general, lymphoma is one of the most common neoplasms in the cat.5,6 In the above retrospective study of 146 cats, 21.9% of cats had neoplasia, which was attributed as the sole case of pericardial effusion in 5.5% of cats, and a possible contributing factor in 7.5% of cats.1 Only 4.8% of cats had a defined mass visualized on echocardiography,1 which is lower than what has been reported in dogs. In a study of 143 dogs with pericardial effusion, a mass was identified on echocardiography in 30.8% of dogs.7 Without staging done in this case, it is impossible to know if the heart was the primary site or if this was a case of metastatic lymphoma with an undiagnosed primary site. There has been one case report of presumptive primary cardiac lymphoma in a cat causing pericardial effusion.8 Unlike the case presented here, the cat in this report had a mass identified on echocardiogram, which was 10 x 7mm and encompassed the left atrioventricular junction.8 Similar to our case, a diagnosis of lymphoma was made on cytologic assessment of the pericardial fluid. However, no other staging tests were performed and a necropsy was not done post-mortem to evaluate for other possible primary sites in the cat of this report.
Although pericardial fluid analysis is often a low yield diagnostic test, this case illustrates an example of its clinical utility. A challenge in interpreting pericardial effusions is that they are almost always hemorrhagic, which complicates the interpretation of nucleated cell counts and total protein. Furthermore, the mere presence of an effusion results in irritation of the mesothelium, and the pericardium is infamous for eliciting mesothelial hyperplasia.9 Cytologically these reactive mesothelial cells are very difficult to differentiate from neoplastic cells.9 In addition, hemangiosarcoma and aortic body tumors, which are the most common heart base tumors in dogs, rarely exfoliate into pericardial effusions.2 But despite the challenges and limited diagnostic value of pericardial fluid analysis, it is the primary means of diagnosing cardiac lymphoma (primary or metastatic).2 In a report of 9 dogs with pericardial effusion attributed to lymphoma, the diagnosis of lymphoma was made on cytological examination of the pericardial fluid in all cases.3 In general for all effusions, fluid analysis is a minimally invasive technique that can provide a diagnosis in some cases or at minimum shed light to the mechanisms for its presence.
Case follow up
The cat was seen on re-check appointments with the Cardiology and Oncology services. Three months after diagnosis, a return of the pericardial effusion was noted and the cat was tachycardic (300 beats per minute). Additional cardiac medication was recommended (diltiazem) to help slow the heart rate, but was declined by the owners. At that time, a full abdominal ultrasound was performed, which revealed enlarged mesenteric lymph nodes and a diffusely enlarged liver. At the most recent re-check a month later (4 months after diagnosis), the catre-presented for respiratory distress. On echocardiogram, a moderate amount of pericardial effusion was present and the left atrium and pulmonary vein were noted to be severely enlarged. The respiratory distress was attributed to left-sided congestive heart failure rather than cardiac tamponade, based on the lack of right atrium collapse. Medical management was pursued (increased dose of furosemide) due to the risk of lacerating the left atrium during pericardiocentesis. At the time of this report, it is believed that the patient is still alive (5 months after diagnosis).
References
- Hall, Daniel J. et al. Pericardial effusion in cats: a retrospective study of clinical findings and outcome in 146 cats. J Vet Intern Med. 2007; 21: 1002-1007.
- Tobias, AH. Pericardial disorders. In: Ettinger SJ, Feldman EC, eds. Textbook of Veterinary Internal Medicine, 6th ed. St. Louis, MO: Elsevier Saunders; 2005.
- MacGregor, J.M. et al. Pericardial lymphoma with pericardial effusion in 9 dogs (1998-2001). J Vet Intern Med. 2003; 17: 398 [abstract].
- Kittleson, MD. Feline myocardial disease. In: Ettinger SJ, Feldman EC, eds. Textbook of Veterinary Internal Medicine, 6th ed. St. Louis, MO: Elsevier Saunders; 2005.
- Kisseberth, WC. Neoplasia of the Heart. In: Withrow, SJ, Vail DM, Page, RL, eds. Withrow & MacEwen’s Small Animal Clinical Oncology, 5th ed. St. Louis, MO: Elsevier Saunders; 2013.
- Vail, DM. Feline Lymphoma and Leukemia. In: Withrow, SJ, Vail DM, Page, RL, eds. Withrow & MacEwen’s Small Animal Clinical Oncology, 5th ed. St. Louis, MO: Elsevier Saunders; 2013.
- Staffod JM, et al. A retrospective study of clinical findings, treatment and outcome in 143 dogs with pericardial effusion. J Small Anim Pract. 2004; 45:546-552.
- Shinohara, N et al. Presumptive primary cardiac lymphoma in a cat causing pericardial effusion. J Vet Cardiol. 2005; 7: 65-69.
- Rebar, AH and CA Thompson. Body cavity fluids. In: Raskin, RE, Meyer, DJ, eds. Canine and Feline Cytology, 2nd ed. St. Louis, MO. Saunders Elsevier; 2010.