Interpretation
Endocrine/neuroendocrine tumor, likely phaeochromocytoma
Explanation
Given the intermittent bouts of severe systemic hypertension during surgery and evidence of vascular invasion, a pheochromocytoma was the top differential diagnosis for the right adrenal mass (Question 1). Although less likely in this case, an adrenal cortical tumor was another differential diagnosis because some dogs with diabetes mellitus have concurrent Cushing’s disease, which may be from a functional adrenal cortical tumor. Concurrent adrenal medullary and cortical tumors have also been reported.1
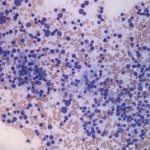
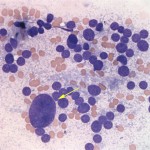
The touch impression smears were of high cellularity and consisted mostly of a monomorphic population of bare nuclei in a pale, slightly granular background from ruptured cytoplasm of the cells. These findings are typical for an endocrine/neuroendocrine tumor, including phaeochromocytoma2 (Question 2). The intact neoplastic cells had round nuclei with coarse chromatin and indistinct nucleoli and moderate amounts of granular blue cytoplasm. Anisocytosis and anisokaryosis were generally mild in most cells with a few cells containing very large nuclei (mild cytologic criteria of malignancy, Question 3). No mitotic figures were noted. The history of hypertension, location, and cytologic features of the adrenal gland mass were most compatible with a malignant pheochromocytoma.
|
|
|
Additional information
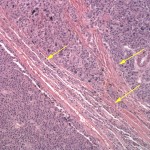
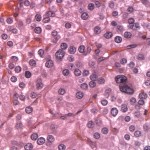
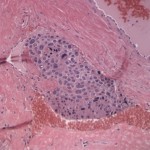
Histopathological examination of the surgical biopsy of the adrenal gland revealed a densely cellular, unencapsulated, and infiltrative neoplasm that extended through the capsule and partially effaced normal adrenal gland architecture (Figure 3). The neoplasm was composed of packets and cords of neoplastic polygonal cell surrounded by fine fibrovascular connective tissue (Figure 4). The morphologic features of the neoplastic cells were similar to those seen on cytologic examination. There was mild to moderate multifocal hemorrhage. Occasionally, aggregates of neoplastic cells were also seen in medium-sized blood vessels (Figure 5). The histopathologic findings are consistent with a malignant pheochromocytoma.
Discussion
Primary adrenal tumors occur in approximately 0.17% to 0.76% of pet dogs (representing 1% to 2% of all canine tumors).1 The clinical presentation of adrenal gland tumors differ depending on whether the tumor is functional or non-functional and if functional, what type of hormone is primarily being secreted. The adrenal cortex is histologically divided into three regions: zona glomerulosa, zona fasciculata, and zona reticularis, which secrete mineralocorticoid (aldosterone), glucocorticoids, and androgens, respectively. The chromaffin cells of the adrenal medulla secrete catecholamines (epinephrine and norepinephrine). Functional adrenal cortical tumors can cause Cushing’s disease due to excess glucocorticoid secretion (the most common functional tumor in dogs) or rarely, Conn’s syndrome with hypokalemia due to excess aldosterone secretion, which has been reported in dogs and cats. In ferrets, many adrenal tumors secrete excess sex steroid hormones, resulting in bilateral alopecia, vulvar enlargement, and/or bone marrow suppression.3
Pheochromocytomas are neoplasms arising from the chromaffin cells of the adrenal medulla and secrete catecholamines, primarily norepinephrine in dogs.1,3 In humans and cattle, pheochromocytomas may develop with concurrent calcitonin secreting C-cell tumor in the parathyroid gland, as part of the multiple endocrine neoplasia (MEN) syndrome.1 Several genetic mutations, such as von Hippel–Lindau (VHL), multiple endocrine neoplasia type 2 (RET), and neurofibromatosis type 1 (NF1), are associated with pheochromocytoma and extra-adrenal paraganglioma in humans.4 Interestingly, approximately half of dogs with pheochromocytomas have other concurrent endocrine (thyroid, adrenal cortex, chemodectoma) or non-endocrine tumors (carcinomas, lymphoma, hemangiosarcoma).5
Pheochromocytomas are uncommon in dogs and accounts for 0.01% to 0.1% of all canine tumors and are most commonly diagnosed in middle-aged to older dogs. No apparent gender or breed predilection has been reported.5,6 Approximately half of pheochromocytomas are found as incidental findings at necropsy or surgery.5,6 The most common clinical signs at presentation are non-specific clinical signs, such as weakness and lethargy and approximately 40% of dogs with pheochromocytomas show cardiopulmonary signs, such as tachyarrhythmia and tachypnea.5 Hypertension, as is seen in this case, has been reported in 43% of dogs with pheochromocytoma.5 No consistent abnormalities in CBC, chemistry panel, or urinalysis have been reported in dogs with pheochromocytoma.5,6 Excess catecholamine secretion could be responsible for the mild hyperglycemia documented on initial biochemical testing n the dog of this report , however it is more likely due to the dog’s underlying diabetes mellitus. The hypercholesterolemia and hypertriglyceridemia were also attributed to the dog’s diabetes mellitus. The mildly increased alkaline phosphatase activity could also be secondary to secretion of endogenous corticosteroids.
Abdominal ultrasonography is the most commonly used imaging procedure for detection of abdominal masses and an adrenal gland mass was identified in 50% of 40 tested dogs with pheochromocytomas in one study.5 Small pheochromocytomas are well capsulated and remain confined to the affected adrenal gland. Malignant pheochromocytomas are often larger and compress or invade into the posterior vena cava, forming a throumbus, as seen in this case.3 Vena caval invasion and distant metastasis are seen in approximately 20-52% and 13-24% of pheochromocytomas, respectively.5,6
Aspiration cytology and ultrasound-or computerized tomography-guided biopsies are not routinely recommended for small adrenal lesions because of the high risk of complications and the inability to reliably differentiate between benign and malignant lesions.1 However, cytology may be useful to differentiate cortical tumors from pheochromocytomas.2 The typical cytologic features of cortical tumors include many intact cells that have round nuclei and markedly vacuolated cytoplasm with a low nuclear cytoplasmic ratio. The vacuolation is consistent with production of lipidogenic hormones. In contrast to cortical tumors, the typical cytologic features of pheochromocytomas include many uniform naked nuclei that are often on a finely granular and basophilic background. Rare intact cells with a high nuclear cytoplasmic ratio can be also seen.2 However, cytologic findings do not always discriminate between these tumors and should not be relied upon. Rather, histopathology and immunohistochemistry with chromogranin A or synaptophysin or both should be performed to confirm a diagnosis of pheochromocytomas in dogs.1,3
In humans, detection of increased concentrations of catecholamines and their metabolites in urine or plasma is the standard test for pheochromocytoma. Measurement of metanephrines in plasma or urine offer the best diagnostic performance and are the tests of first choice for biochemical screening.4 In a study with 7 dogs with pheochromocytoma, a urinary normetanephrine-to-creatinine ratio was significantly increased in dogs with pheochromocytoma compared with dogs with hyperadrenocorticism and healthy dogs.7 In 8 dogs with pheochromocytoma, measurement of plasma-free normetanephrine by liquid chromatography-tandem mass spectrometry showed excellent sensitivity and specificity for a diagnosis of pheochromocytoma.8 However, these measurements are currently not widely available in veterinary medicine.
Surgery is the only definitive treatment for pheochromocytoma in dogs. Treatment with phenoxybenzamine, a noncompetitive alpha-adrenergic agonist, before adrenalectomy substantially decreases perioperative mortality in dogs with pheochromocytomas.3,9
Follow-up
The dog was returned to the hospital for recheck 6 weeks after the surgery and was doing well. It was then lost to follow-up.
References
- Capen, C. Tumors of Endocrine Glands. 2002. In: Tumors of Domestic Animals. Meuten D. 4th ed, pp. 607-696. Iowa State Press.
- Bertazzolo, W., Dider, M., and Gelain, M.E. 2014. Accuracy of cytology in distinguishing adrenocortical tumors from pheochromocytoma in companion animals. Vet Clin Path 43, 453-459.
- Bailey, D.B. and Page, R.L., Tumors of the Endocrine System. 2013. In: Withrow and Macewen’s Small Animal Clinical Oncology. Withrow SJ, Vail DM, Page R. 5th ed, pp. 583-609. Elsevier, St. Louis, MO.
- Berkel, A., Lenders, J.W., and Timmers, H.J. 2014. Biochemical diagnosis of phaeochromocytoma and paraganglioma. Euro J Endocri 170, 109-119. Was 7
- Barthez, P.Y., Marks, S.L., Woo, J., et al. 1997. Pheochromocytoma in dogs: 61 cases (1984-1995). J Vet Intern Med 11, 272-278.
- Gilson, S.D., Withrow, S.J., Wheeler, S.L., et al. 1994. Pheochromocytoma in 50 dogs. J Vet Intern Med 8, 228-232.
- Quante, S., Boretti, F.S., Kook, P.H. 2010. Urinary catecholamine and metanephrine to creatinine ratios in dogs with hyperadrenocorticism or pheochromocytoma, and in healthy dogs. J Vet Intern Med 24, 1093–1097.
- Gostelow, R., Brindger, N., and Syme, H.M. 2013. Plasma-free metanephrine and free norepinephrine measurement for the diagnosis of pheochromocytoma in dogs. J Vet Intern Med 27, 83-90.
- Kyles, A.E., Feldman, E.C., De Cock, H.E.V., et al. 2003. Surgical management of adrenal gland tumors with and without associated tumor thrombi in dogs: 40 cases. J Am Vet Med Assoc 223, 654-662.
Authors: M. Asakawa (clinical pathology resident), T. Stokol