Interpretation
Extravascular hemolytic anemia due to hemotropic Mycoplasma infection (Mycoplasma haemofelis) with a secondary immune-mediated hemolytic anemia (intra- and extravascular hemolysis)
Explanation
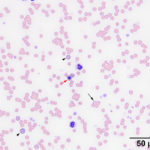
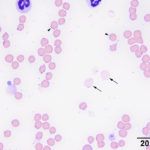
Many Mycoplasma organisms, with their characteristic epicellular location and rod, ring-like, and coccoid shapes, were adhered to red blood cells (Figures 1-2A, Question 3). Their large size is compatible with Mycoplasma haemofelis. Speciation can be confirmed using specific primers and polymerase chain reaction (PCR) testing for parasitic DNA (see below). Pertinent red blood cell morphologic changes in the smear included mild polychromasia (absolute reticulocyte count 30.8 thou/μL, reference interval 8.5-60.7 thou/μL), supporting a beginning regenerative response. There was also erythrocyte agglutination and many spherocytes, which indicated a secondary immune-mediated hemolytic anemia, primarily due to extravascular hemolysis (Questions 1 and 2). Spherocytes are thought to be formed from trogocytosis, when macrophages remove portions of cells bearing adhered immunoglobulin via Fc receptors, however antibody- or complement-induced changes in the lipid bilayer of the red blood cell membrane could also be responsible. Spherocytes can be difficult to conclusively identify in cats because their red blood cells normally lack central pallor. Agglutination results when high concentrations of IgG antibody or multimeric IgM antibody crosslinks adjacent red blood cells, forming three-dimensional aggregates. There were also mild to moderate numbers of ghost cells, a highly unusual finding in cats with this erythroparasite (Figure 2aA. Ghost cells, per se, indicate rupturing of red blood cells, leaving a remnant of membrane (“ghost” of itself). These are usually an artifact of collection, storage or smear preparation (in vitro hemolysis), but their large numbers in a freshly collected blood sample, together with the mildly hemolyzed plasma, support true in vivo intravascular hemolysis in this case (Questions 1 and 2). The latter is likely due to the assembly of the membrane-attack-complex of complement on red blood cell membranes, with complement being activated through the classical (immunoglobulin-mediated) or alternate (damage- or pathogen-associated molecular pattern-mediated pathways.
|
|
|
Discussion
Hemotropic Mycoplasma spp. are small (0.3-0.8 µm) gram positive and non-acid-fast bacterium that lack a cell wall.1,2 They can infect many different species and are distributed worldwide.1,2 These epicellular organisms attach to and replicate on the surface of red blood cells and can be seen as cocci-, rod-, or ring-shaped organisms on red blood cells during blood smear evaluation.1,2 Mycoplasma spp. have evolved with a very small genome that makes them rely on the host to supply metabolites and nutrients.3 They reproduce by binary fission and contain both DNA and RNA.1 Even with their reduction in genome size, Mycoplasma spp. retain vital genes, such as those encoding lipoproteins and membrane-binding proteins, that are important to their pathogenesis and virulence.3 Originally these hemotropic Mycoplasma spp. were classified in the order Rickettaiales, family Anaplasmataceae, and genera Haemobartonella and Eperythrozoon.1,2,4 Through phylogenetic and sequencing analysis of the 16S rRNA, these organisms have been reclassified as Mycoplasma spp.1,2,4
There are three Mycoplasma spp. that have been shown to infect cats: Mycoplasma haemofelis, Candidatus Mycoplasma haemominutum, and Candidatus Mycoplasma turicensis.1,2 The “Candidatus” term is given to newly discovered hemotropic Mycoplasma spp. where there is insufficient evidence to support their speciation.2 These organisms vary in their pathogenicity and the severity of disease they cause.2 Mycoplasma haemofelis is the least prevalent but the most pathogenic amongst these three organisms.1,2 They are larger and more readily seen on blood smear evaluations and can cause a moderate to marked anemia, or the disease coined as feline infectious anemia.1,2 The anemia is thought to be partly mediated by direct damage to the erythrocyte by the organism, but the main cause appears to be related to secondary immune-mediated destruction of the erythrocytes.1,2 Cold and warm erythrocyte-bound antibodies have been shown to appear at different times after experimental infection.1 Extravascular hemolysis is the primary mechanism of the anemia but there have been rare reports of an intravascular component in some infected cats.2 Concurrent infections with Feline Leukemia Virus (FeLV) and/or Feline Immunodeficiency Virus (FIV) have been reported to produce a more severe anemia.1,2 Candidatus Mycoplasma haemominutum appears to be more prevalent but much less pathogenic, than Mycoplasma haemofelis.1,2 This organism is smaller in size and more difficult to identify in a blood smear.1,2 Most often an infection with this organism produces no clinical signs or noticeable anemia.1,2 However, immunosuppression or concurrent infection with FeLV or FIV may elicit an anemia in cats with Candidatus Mycoplasma haemominutum.1,2 Candidatus Mycoplasma turicensis is a newer discovered organism that was initially isolated from a Swiss cat population and has also been found in South African, United Kingdom, and Australian cats.5 The prevalence of the latter is low, similar to Mycoplasma haemofelis, and it can cause a mild anemia when there are other cofactors, such as co-infection with another Mycoplasma spp. or immunosuppression.5 Many theories exist as to how transmission of these organisms occur. Fleas and ticks have been studied as possible vectors but the evidence for this is minimal.2,6,7 One study was able to isolate Mycoplasma DNA from fleas that fed on an infected cat but only 1 out of 8 cats became transiently infected after being exposed to fleas carrying Mycoplasma spp, indicating an inefficient mode of transmission.6 Aggressive behavior and direct inoculation through bite wounds has also been studied.2 There appears to be a strong association between bite wound abscesses preceding infections with Mycoplasma spp.1,2 Other risk factors commonly seen with Mycoplasma infections include male sex, outdoor exposure, and concurrent retroviral infection, all of which would support the hypothesis of direct bite wound inoculation.2,7 Studies have suggested that more aggressive activities that elicit bleeding need to be present to cause infection, since organism levels in saliva have been shown to be low and cats that have ingested infected blood have developed Mycoplasma infection.2
The clinical symptoms associated with a hemotropic Mycoplasma infection depend on the severity of the anemia. A mild anemia may result in lethargy, mild mucous membrane pallor, and some inappetence.2 If the anemia is severe, patients can experience marked lethargy to obtundation, marked mucous membrane pallor, anorexia, tachycardia, a physiologic murmur, and tachypnea. Affected animals can be hypothermic or febrile.2 On laboratory testing, the anemia is usually regenerative and increased Howell-jolly bodies may be seen. Rare cases may show a prominent metarubricytosis. Agglutination can occur in some but not all infected cats.2 Ghost cells, as seen in this case, indicate concurrent intravascular hemolysis and are produced when the red blood cell membrane is damaged by antibody-mediated complement fixation, but this is a rare finding with this erythroparasite.1 If a regenerative anemia is not noted, the patient should be tested for FeLV to rule out a concurrent infection (assuming it is not a peracute onset anemia).2 The white blood cell count may be normal, increased or low depending on co-morbidities.2
Many attempts have been made to culture these fastidious Mycoplasma spp. all of which have proven to be unsuccessful to date.1,2,7 A definitive diagnosis of Mycoplasma spp. can be made on blood smear examination for M. haemofelis particularly (the other species are really small and hard to conclusively identify). However, organisms are only visible in about 50% of cases during the acute phase of the disease. Stain precipitate or water artifact can be readily misidentified as these organisms, however the organisms tend to indent the red blood cell membrane slightly (they are epicellular) and are in the same plane of focus as the red blood cells, whereas stain precipitate is more variably sized, often in a different plane of focus and does not affect the cell outline. Water artifact is refractile if you focus up and down. A pretreatment blood smear should be examined because the organisms can rapidly disappear after initiation of therapy.1,2 Evaluation of a freshly drawn blood sample is recommended since these organisms can detach from the red blood cells when stored in ethylenediamine tetraacetic acid (EDTA).1,2 Genetic testing with PCR analysis has become the gold standard for identifying Mycoplasma infections due to its high sensitivity and is recommended if a blood smear examination is negative or inconclusive.1,2,7 Originally, Mycoplasma infections where diagnosed using conventional PCR assays, which detect bands at or around specific base pair sizes on an agarose gel.2,7 Advancements in PCR analysis has allowed for the development of real-time PCR assays.7 Real-time PCR not only can detect Mycoplasma organisms but can also identify the specific species by using specific probes with fluorometric detection.2,7 This type of assay has reduced false positive results but false negative results can still occur if the available probes are not specific for a select species.2,7
Treatment of Mycoplasma infection depends on the severity of the clinical symptoms and requires administration of an antibiotic for 3-4 weeks.1 For patients that are not anemic and PCR positive, treatment is not recommended since antibiotics are not innocuous and no treatment has been shown to completely eliminate the organism.2 Doxycycline (10 mg/kg/day), given for 3 weeks, has long been the treatment of choice for Mycoplasma infections.1,2,8 There has been ongoing research to find alternative treatments since doxycycline can cause gastrointestinal disease and esophagitis with stricture formation in some cats.1,2 Evidence shows that fluoroquinolones, such as enrofloxacin and marbofloxacin, have good efficacy against Mycoplasma spp. and may clear the organism for a longer period.1,2,8 The fuoroquinolone Pradofloxacin has proven to be effective against Mycoplasma haemofelis and clears the organism for longer than doxycycline.1,8 Other treatments besides antibiotics include blood transfusions for severely anemic patients, intravenous fluid therapy with supportive care and glucocorticoids to reduce immune-mediated red blood cell destruction, although the latter treatment option is controversial.1,2
There have been reports of hemotropic Mycoplasma in immunocompromised human patients.1 According to a review, one case involved an immunodeficient patient in Brazil that was infected with Bartonella henselae and Mycoplasma haemofelis.1 Another article found co-infections of Anaplasma platys, Bartonella henselae, and Candidatus Mycoplasma haematoparvum in a young veterinarian working with animals in Grenada, Ireland, and South Africa.9 These findings suggest that Mycoplasma spp. could be zoonotic and caution should be used when handling samples from infected animals.1,2
References
- Messick, JB. and Harvey, JW. Hemotropic Mycoplasmosis (Hemobartonellosis). In: Green C.E., Infectious Disease of the Dog and Cat, Fourth edn. St. Louis, Elsevier Saunders; 2012. P. 310-319.
- Sykes, J.E. Feline Hemotropic Mycoplasmas. Veterinary Clinics: Small Animal Practice 2010, 40. 1157-1170.
- Santos, A.P., Guimaraes, A., Nascimento, N.C., SanMiguel, P.J., Martin, S.W., Messick, J.B. Genome of Mycoplasma haemofelis, unraveling its strategies for survival and persistence. Veterinary Research 2011, 42: 102.
- Neimark, H., Johansson, K-E., Rikihisa, Y., Tully, J.G. Proposal to transfer some members of the general Haemobartonella and Eperythrozoon to the genus Mycoplasma with descriptions of ‘Candidatus Mycoplasma haemofelis’, ‘Candidatus Mycoplasma haemomuris’, ‘Candidatus Mycoplasma haemosuis’, and ‘Candidatus Mycoplasma wenyonii’. International Journal of Systematic and Evolutionary Microbiology 2001, 51: 891-899.
- Willi, B., Tasker, S., Boretti, F.S., et al. Phylogenetic Analysis of “Candidatus Mycoplasma turicensis” Isolates from Pet Cats in the United Kingdom, Australia, and South Africa, with Analysis of Risk Factors for Infection. Journal of Clinical Microbiology 2006, 44: 4430-4435.
- Woods, J.E., Brewer, M.M., Hawley, J.R., Wisnewski, N., Lappin, M.R. Evaluation of experimental transmission of Candidatus Mycoplasma haemominutum and Mycoplasma haemofelis by Ctenocephalides felis to cats. American Journal of Veterinary Research 2005, 66: 1008-1012.
- Sykes, J.E., Drazenovich, N.L., Ball, L.M., Leutenegger, C.M. Use of Conventional and Real Time Polymerase Chain Reaction to Determine the Epidemiology of Hemopasma Infections in Anemic and Nonanemic Cats. Journal of Veterinary Internal Medicine 2007, 21: 685-693.
- Dowers, K.L., Tasker, S. Radecki, S.V., Lappin, M.R. Use of pradofloxacin to treat experimentally induced Mycoplasma hemofelis infection in cats. American Journal of Veterinary Research 2009, 70: 105-111.
- Maggi, R.G., Mascarelli, P.E., Havenga, L.N., Naidoo, V., Breitschwerdt, E.B. Co-infection with Anaplasma platys, Bartonella henselae, and Candidatus Mycoplasma haematoparvum in a veterinarian. Parasites and Vectors 2013, 6: 103.
Authored by: S. Chu (Clinical Pathology Intern) and edited by T. Stokol