Interpretation
Hematopoietic neoplasia, either lymphoma with a leukemic phase or a primary leukemia
Explanation
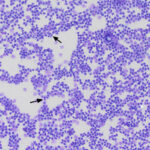
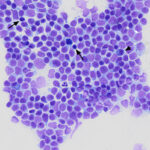
Blood smear: Low numbers of intermediate to large cells were identified in the initial blood smear in June. At that time, they were “flagged” by the clinical pathologist reviewing the smear as “other WBC” with a comment that they could be reactive or neoplastic. However, there were increasing numbers of these cells with subsequent visits (on the first, second and third hemograms, the cells represented 3%, 5% and 17% of a 100-cell differential count, respectively), allowing more detailed assessment of their features. The cells were relatively uniform in appearance. They had convoluted nuclei and small amounts of light to medium blue cytoplasm, which contained low numbers (1-3) of small discrete margined vacuoles. Nuclear chromatin was fine, with 1-2 indistinct nucleoli (Figures 1-2). Their uniform features supported a hematopoietic neoplasm, either leukemic phase of lymphoma or a primary leukemia, versus reactive lymphocytes. In addition, the fine chromatin pattern (mostly euchromatin) and presence of nucleoli in the cells were more compatible with an immature cell (blasts), i.e. leukemic phase of intermediate to large lymphoma or an acute leukemia (of myeloid or lymphoid origin) over a more indolent neoplasm. Given the lack of cytopenias on the first visit and relatively mild cytopenias that developed over time, a leukemic phase of lymphoma or stage V lymphoma was favored over an acute leukemia (Question 1).
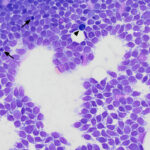
Prescapular and popliteal lymph node aspirates: The smears of the aspirates contained many intermediate to large cells resembling those seen in blood. There were low numbers of residual small lymphocytes, plasma cells and large residual lymphocytes with dark blue cytoplasm. Mitotic figures were seen in the neoplastic population (Figures 3-7). Given the bicytopenia was mild and a recent development, the cytologic diagnosis was lymphoma. Although the cytoplasmic tailing of the cells in the lymph node smears mimicked that seen in tumor cells from indolent T zone lymphoma (which can have a leukemic phase, i.e. tumor cells in circulation),1 the tumor cells had other cytologic features that were inconsistent with T zone cells (finer chromatin, multiple nucleoli, deeply convoluted nuclei and less cytoplasm). The persistent thrombocytopenia and progressive non-regenerative anemia are also uncommon hematologic findings with indolent disease.
Additional tests
An aspirate of the prescapular lymph node was sent to Colorado State University for flow cytometric-based immunophenotyping to further characterize the origin of the neoplastic cells (Question 2). The majority of the intermediate to large cells were only positive for the hematopoietic stem cell marker, CD34, and were negative for major histocompatibility complex class II (MHCII), B cell (CD21, CD 22), T cell (CD3, CD5) and myeloid (CD4, CD14, CD18) antigens. The results indicated an acute leukemia that could not be characterized further as myeloid or lymphoid (“unclassified” acute leukemia).
Outcome
The patient was administered CCNU (chosen for its ability to traverse the blood-brain barrier) in tandem with prednisolone as chemotherapy, as well as levetiracetam for seizure control. CCNU was initially well-tolerated and partial remission was achieved, however following three treatments, the CCNU had to be discontinued due to a progressive hepatopathy (increased transaminase, alkaline phosphatase and gamma-glutamyl transferase activities). The patient was switched to alternating doses of doxorubicin and cytarabine and administered a liver support supplement. Unfortunately, following two treatments of doxorubicin and one of cytarabine, the patient acutely declined, presenting to CUHA’s emergency service with lethargy and vomiting. Pneumonia was identified on imaging during hospitalization and blood work showed pancytopenia with an inflammatory leukogram (left shift and toxic change). Euthanasia was elected due to a poor clinical picture and presumptive sepsis, secondary to the pneumonia. The dog had a total survival time of 195 days from the first incidence of neoplastic cells noted on blood smear examination and 117 days from diagnosis. Notably, prior to this decline, the patient was largely clinically well, with no further seizure episodes, and minimal difficulties reported at home.
Discussion
Leukemia at its most base definition is a proliferation of neoplastic cells in bone marrow at various points in the hematopoietic tree of development and can be of myeloid or lymphoid origin or acute or chronic. A primary leukemia is contrasted with a primary lymphoid tumor in the periphery, which has concurrent circulating cells, also known as stage V lymphoma (we use the term leukemic phase of lymphoma for this scenario). However, in individual animals with neoplasms, it can be difficult to distinguish between a primary leukemia (of any origin) and lymphoma, as illustrated in this case. Chronic leukemias consist of an expansion of more mature or differentiated cells and usually have a more indolent course (particularly chronic lymphocytic leukemia). In contrast, acute leukemias usually consist of a proliferation of more immature cells, which efface the bone marrow, resulting in bi- or pancytopenia, and usually have a rapid clinical progression. In humans, extensive immunologic probes are applied to aid in differentiating hematopoietic neoplasms by lineage (e.g., lymphoid or myeloid) and now genetic testing is included to further classify the disease on the basis of driver mutations or gene fusions, allowing a fine degree of granularity in diagnosis.2,3 In contrast, in veterinary medicine there is a much more limited array of probes and genetic testing has just begun.4 To yield a definitive diagnosis of acute leukemia, we typically rely on morphologic features implying cell immaturity (fine chromatin or nucleoli or both) in 20% or more of cells (i.e. blasts) in blood or (ideally) bone marrow and expression of the stem cell marker, CD34, with concurrent lack of MHCII on flow cytometric analysis (note that not all types of acute leukemia will express CD34 and myelomonocytic variants of acute myeloid leukemia [AML] can express MHCII). It is not completely out of the norm to have a leukemia in a dog which defies classification beyond acute leukemia, as with this case.4,5 Such cases have been referred to as acute undifferentiated leukemia,5-7 however because we apply a limited repertoire of antibodies to phenotype the leukemia, we prefer the term acute lineage negative leukemia (ALNL). In prior reports, these uncategorizable leukemias comprised 18-24% of cases.4,5 In a recently concluded study from our institution, we found that of 108 dogs with acute leukemia, 19% were ALNL.
Both AML and acute lymphoblastic leukemia (ALL) tend to be diseases of middle-aged dogs, with a male predominance, and increased representation of German shepherd dogs, Labrador retrievers, and Golden retrievers.4,5,7-9 In our recent study, the median ages were 7, 5 and 8.5 years and percentage of intact or neutered males was 60, 62 and 75% for dogs with AML, ALL, and ALNL, respectively. The most common purebred dogs were Golden and Labrador retrievers followed by German shepherds, which were over-represented based on commonality of the breed. Presenting clinical signs and physical examination findings in acute leukemia tend to be nonspecific but animals are usually sick. Three cases series of acute leukemia found the most commonly reported clinical signs to be lethargy and inappetence.7,9-10 Two case series did report a low, but comparable, number of cases with neurologic abnormalities (around 16-17% ),7,9 as seen in this case, which had the primary complaint of seizures. Otherwise the dog from this case was well, showing no other clinical signs or physical examination findings associated with acute leukemia at the first and second visits. It was only at the third visit that mild peripheral lymphadenopathy was noted, which was several weeks after the first documented seizure.
Clinicopathologic findings on diagnosis of an acute leukemia most often include bi- or pancytopenia,7,9-10 which is attributed primarily to myelophthisis. Biochemical results are frequently not informative, consisting of high alkaline phosphatase activity (which could be due to excessive endogenous corticosteroids)7,10 and hyperglobulinemia and low albumin concentrations, which may reflect concurrent inflammation.10 However, affected animals may have no cytopenias, as seen in this case initially, which has been reported in 9-11% of dogs in two studies.9-10 A third of cases in one study of AML in dogs had a leukocyte count within reference intervals, as seen with our case, although the remaining dogs had a leukocytosis.9
CD34, a sialomucin found on hematopoietic stem cells, is generally considered to be a marker of an acute leukemias when found on circulating cells that also lack MHCII expression.11 Lymphomas, usually of B cell origin, can express CD34, but usually concurrently express MHCII.12 This case had a predominance of large cells in aspirates of the prescapular lymph node on flow cytometric analysis that were expressing CD34 and lacked typical differentiation markers for lymphoid or myeloid cells, implying that the leukemia originated from an early progenitor cell, which expanded clonally. The cells also appeared immature and lacked features of myeloid differentiation (e.g. monocytoid nuclei) on morphologic assessment.13 Now that single cell RNA sequencing has been performed on normal canine bone marrow,14 it will be possible to use this data to assign a “cell of origin” signature to cases of acute leukemia. Unfortunately, RNA sequencing was not done on this dog’s tumor.
Treatment for acute leukemias is often a decision of aggressive versus palliative treatment versus euthanasia. Aggressive treatment most commonly consists of single agent (e.g., L-asparaginase) versus multiagent chemotherapy (e.g., CHOP [cyclophosphamide, doxorubicin, vincristine, prednisone]), although doxorubicin-cytarabine-based protocols are used by some clinicians to treat AML.9 Median survival times are usually measured in weeks.4,7,9-10 Survival time is also short in CD34-positive T cell leukemias with a reported median survival time of 16 days in 12 dogs, nine of which received CHOP therapy.15 It is not known if dogs with ALNL have poorer survival versus dogs with phenotyped AML or ALL.
This case contained a number of deviations from expected findings with an acute leukemia. The patient was clinically healthy, apart from approximately one seizure per month with an unrelated comorbidity of presumptive hip osteoarthritis. The dog remained healthy for an additional four months after diagnosis of an acute leukemia, which is a long survival time for this disease. The patient’s initial hemogram lacked cytopenias and low numbers of concerning cells were only uncovered on blood smear examination. Tumor cells were only conclusively identified in the hemogram on the second and third visits. The initial cytologic diagnosis on smears from lymph node aspirates was lymphoma versus the more generic term, hematopoietic neoplasia, because the dog only had low numbers of cytopenias and circulating tumor cells. In addition, the dog had mild peripheral lymphadenopathy, which was only detected six weeks after initial observation of concerning cells in the circulation. The relative lack of cytopenias suggests that this dog had primary extramedullary disease, as has been reported in dogs with AML, with the tumor primarily being located in the lymph node and not the bone marrow.10 However, a bone marrow aspirate was declined by the owner and we cannot exclude the possibility that the acute leukemia would have been present in the bone marrow and able to be diagnosed from a bone marrow aspirate. It is likely that this dog would have developed an overt leukemia, even with the ongoing chemotherapy, similar to other cases of “isolated extramedullary” AML we have seen.10 This case also illustrates the value of a blood smear examination, versus relying on numerical or automated generated data.
Author: Kate Marciano (resident), edited by T. Stokol
References
- Seelig DM, Avery P, Webb T, Yoshimoto J, Bromberek J, Ehrhart EJ, et al. Canine T‐Zone Lymphoma: Unique Immunophenotypic Features, Outcome, and Population Characteristics. J Vet Intern Med. 2014;28(3):878–86.
- Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IB de O, Berti E, et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia. 2022 Jul;36(7):1720–48.
- Khoury JD, Solary E, Abla O, Akkari Y, Alaggio R, Apperley JF, et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia. 2022 Jul;36(7):1703–19.
- Harris R, Bienzle D, Meichner K, Springer N, Glaubitz J, Stokol T. Dogs with acute leukemia have shared and unique genetic mutations compared to human acute leukemia. Blood. 2025 Nov 3;146(Supplement 1):3500–3500 (abstract).
- Novacco M, Comazzi S, Marconato L, Cozzi M, Stefanello D, Aresu L, et al. Prognostic factors in canine acute leukaemias: a retrospective study. Vet Comp Oncol. 2016;14(4):409–16.
- Qasrawi A, Victor G, Munker R. Acute Undifferentiated Leukemia. Blood. 2019 Nov 13;134:3847.
- Bennett AL, Williams LE, Ferguson MW, Hauck ML, Suter SE, Lanier CB, et al. Canine acute leukaemia: 50 cases (1989–2014). Vet Comp Oncol. 2017;15(3):1101–14.
- Avery AC. The Genetic and Molecular Basis for Canine Models of Human Leukemia and Lymphoma. Front Oncol [Internet]. 2020 Jan 24 [cited 2025 Nov 11];10. Available from: https://www.frontiersin.org/journals/oncology/articles/10.3389/fonc.2020.00023/full
- Davis LL, Hume KR, Stokol T. A retrospective review of acute myeloid leukaemia in 35 dogs diagnosed by a combination of morphologic findings, flow cytometric immunophenotyping and cytochemical staining results (2007-2015). Vet Comp Oncol. 2018;16(2):268–75.
- Kaur J, Diamantino G, Morrison K, Meichner K, Springer NL, Hoffman M, et al. Acute myeloid leukemia with peripheral lymph node involvement in dogs: A retrospective study of 23 cases. Vet Pathol. 2025 Mar 1;62(2):195–205.
- Harris RA, Rout ED, Yoshimoto JA, Avery PR, Avery AC. Using digital RNA counting to establish flow cytometry diagnostic criteria for subtypes of CD34+ canine acute leukaemia. Vet Comp Oncol. 2022 Sept, 20(3): 710-719.
- Rao S, Lana S, Eickhoff J, Marcus E, Avery P r., Morley P s., et al. Class II Major Histocompatibility Complex Expression and Cell Size Independently Predict Survival in Canine B-Cell Lymphoma. J Vet Intern Med. 2011;25(5):1097–105.
- Stokol T, Thomas SI, Hoffman M, Zhao S. Flow cytometric-based detection of CD80 is a useful diagnostic marker of acute myeloid leukemia in dogs. Front Vet Sci. 2024 Aug 19;11:1405297.
- Ammons DT, Contursi C, Olsen M, Yoshimoto JA, Owens E, Harris M, et al. Transcriptional landscape of canine hematopoiesis and cross-species comparisons revealed by single-cell RNA sequencing [Internet]. Research Square; 2025 [cited 2026 Feb 5]. Available from: https://www.researchsquare.com/article/rs-6299609/v2
- Williams MJ, Avery AC, Lana SE, Hillers KR, Bachand AM, Avery PR. Canine Lymphoproliferative Disease Characterized by Lymphocytosis: Immunophenotypic Markers of Prognosis. J Vet Intern Med. 2008;22(3):596–601.