Pancytopenia is defined as anemia (typically non-regenerative), neutropenia, and thrombocytopenia. The term implies that the bone marrow is responsible for the cytopenias, with very few extramedullary diseases producing this constellation of hematologic abnormalities. Exceptions are severe bacterial sepsis which can result in anemia of inflammatory disease, severe neutropenia with a left shift and toxic change from inflammation, and thrombocytopenia due to concurrent endotoxemia (sequestration) or disseminated intravascular coagulation (consumption). Cytauxzoon felis and feline infectious peritonitis can also result in such hemogram changes, likely due to similar reasons.
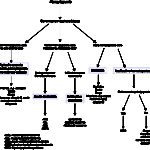
When pancytopenia is present in an animal, the first thing most people think of is that the bone marrow will be aplastic and the prognosis is poor. However, there are several different mechanisms for pancytopenia, a few of which do not have a poor prognosis (such as immune-mediated ineffective marrows). These mechanisms (and causes) only be distinguished by aspiration of the bone marrow.
- Bone marrow aplasia: This is characterized by a lack of hematopoietic cells in the bone marrow, leaving only fat and stromal cells (lymphocytes, plasma cells, mast cells, histiocytes, fibroblasts) and occurs due to destruction or suppression of early multipotential hematopoietic stem cells. This can be due to bracken fern poisoning (cattle), drugs (chloramphenicol, griseofulvin, trimethroprim sulfonamides, chemotherapeutic agents, albendazole, fenbendazole), hormones (estrogen), immune-mediated disease, infectious organisms (chronic Ehrlichia canis) and mycotoxins (Glanemann et al 2023). A syndrome of bovine neonatal pancytopenia has been identified in calves that present with clinical signs of hemorrhage. This is attributed to immune-mediated destruction of precursors in marrow as a consequence of bovine virus diarrhea vaccination of dams, with calves receiving offending allo-antibodies from colostrum. Although the incidence of clinical disease is low, subclinically affected calves with abnormal hemograms can be detected in herds with obvious clinical cases (Bell et al 2014). Selective absence of one cell line with normal production of cells of the other two is called pure red cell aplasia, pure white cell aplasia, or amegakaryocytosis. Immune-mediated attack on committed progenitor cells of the affected series is the apparent cause in most cases.
- Bone marrow hypoplasia: This is characterized by decreased but not absent hematopoietic cells. It can be due to the same causes as bone marrow aplasia.
- Bone marrow necrosis: Uncommon; rarely a cause of pancytopenia. This can be due to ischemic injury (vasculitis), myelotoxins (drugs), viruses (e.g. canine parvovirus), radiation, and drugs. Core biopsy is generally considered more reliable for diagnosis than an aspirate, but we have identified bone marrow necrosis on aspirates.
- Ineffective hematopoiesis: This is associated with hyperplasia of cell lines in bone marrow but peripheral cytopenias. This can be due to bone marrow neoplasia or non-neoplastic conditions.
- Neoplasia: Myelodysplastic syndrome, a clonal hematopoietic neoplasm (also called primary dysmyelopoiesis), will result in cytopenias (solitary, bi- or pancytopenia). Cytopenias are persistent, non-regenerative or non-responsive, and are accompanied by a hyperplastic marrow with morphologic evidence of dysplasia (abnormal production) in some or all lines. Histiocytic sarcoma is another cause of ineffective hematopoiesis, likely due to hemophagocytosis by the tumor cells, although alterations in the marrow microenvironment or immune-mediated destruction is possible.
- Non-neoplastic conditions: These can result in pancytopenia with a hypercellular marrow. Mechanisms include immune-mediated destruction of marrow progenitors (as thought to occur in the precursor-directed or non-regenerative immune-mediated anemias), drug-induced injury (e.g. cephalosporin, phenobarbital, azathioprine, vincristine; this can also be immune-mediated), mycotoxins, and infectious organisms (Ehrlichia canis). An altered marrow microenvironment from drug or toxic injury can result in pancytopenia with ineffective hematopoiesis. Note that mild dysplasia can be seen in these conditions and does not mean that the cytopenias are due to myelodysplastic syndrome.
- Bone marrow neoplasia: This can be due to hematopoietic neoplasms arising in the bone marrow and effacing the marrow due to a growth advantage (e.g. acute myeloid leukemia) or infiltrating neoplasms (e.g. multiple myeloma, histiocytic sarcoma, lymphoma). Acute leukemias are usually (but not always) characterized by cytopenias, which affect two or more lineages. Infiltrating neoplasms can cause cytopenias by extensive infiltrates, which crowd out marrow cells (myelophthisis) or, with fewer infiltrates, an altered microenvironment, usage of vital nutrients depriving normal marrow cells , or through immune-or cytokine-mediated destruction or suppression of hematopoiesis. Similarly, acute myeloid or lymphoid leukemia may not always efface the marrow but still alter the marrow environment causing cytopenias. Some tumors are not always evident on initial marrow aspirates and may only be diagnosed when the animal fails to respond to immunosuppressive therapy. This has been our experience with histiocytic sarcomas in particular, which may not always be seen in marrow in sufficient numbers or with sufficient cytologic criteria of malignancy to confirm neoplasia.
Myelofibrosis may be seen with many of these causes and is a secondary phenomenon that may contribute to but will not be the primary cause of the cytopenias.
