Interpretation
Hepatic coccidiosis
Explanation
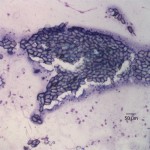
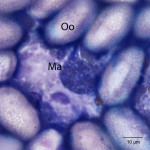
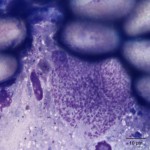
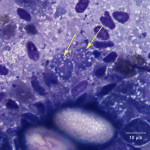
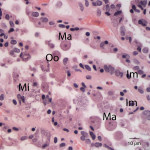
The touch impression smear of the liver revealed individual to large aggregates of numerous, extracellular, oval, clear structures (Figure 1 and 2) on a granular, pale background. In addition, low numbers of round oval structures containing numerous 1-2um, basophilic internal structures (Figure 3) and smaller, round basophilic structures (Figure 4) were also noted. Low numbers of cuboidal epithelial cells (presumptive biliary epithelial cells), ruptured cells, rare heterophils and small lymphocytes were identified (not shown). The structures represent various stages of apicomplexan protozoa. Clear to pale oval unsporulated oocysts, which were 10-20 um x 30-50 um in size and slightly refractile, with thick walls and lightly pink, granular internal structures, dominated (Question 1; Figures 1-2). There were fewer maturing macrogametes, intact and ruptured microgametes, and low numbers of immature gametocytes. Maturing macrogametes were round to oval and contained lightly eosinophilic and basophilic granules (Figure 2). Microgamonts were round and 15-30 um in diameter and were characterized by several to numerous (>100) small basophilic internal nuclei (Figure 3). Undifferentiated, immature gametocytes were round, 8- 15 um in diameter, and have round pale nuclei and basophilic, slightly vacuolated cytoplasm (Figure 4).
|
|
Additional tests
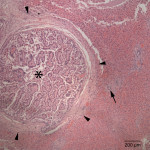
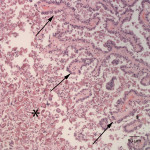
Histopathological examination of the liver showed severe, multifocal, chronic proliferative and lymphoplasmacytic cholangiohepatitis with intraepithelial and intraluminal protozoa. The bile ducts of the liver were multifocally and markedly dilated up to 3 mm in diameter and compressed the adjacent hepatic parenchyma (Figure 5). The bile ducts were lined by hyperplastic biliary epithelium forming multiple papillary projections into the lumen (Figure 6). The bile ducts were surrounded by variably thick fibrous connective tissue (fibrosis). Many biliary epithelial cells and the biliary lumen contained protozoal macrogametes and microgametes in various stages of gametogony (Figure 7). The macrogametes were round, 20-40 um in diameter, and characterized by a central round nucleus with a prominent nucleolus and lightly eosinophilic granular cytoplasm with occasional brightly eosinophilic, peripheral granules. The microgametes were round, 15-30 um in diameter, with lightly basophilic granules. The unsporulated oocysts were oval in shape, 20-40 um in diameter with thick refractile walls that were often collapsed. There was multifocal, mild inflammatory infiltrates composed of lymphocytes, plasma cells, and low numbers of scattered heterophils in periportal areas. The morphologic features of the organisms and histopathological lesions were characteristic for Eimeria stiedae (Question 2).
Discussion
Eimeria stiedae, a coccidian in the protozoal phylum Apicomplexa, parasitizes bile duct epithelium in rabbits and is an important cause of mortality in commercial rabbit farms, especially in weanling rabbits.1,2,5 As many as 16 species of Eimeria have been described in rabbits, of which Eimeria stiedae is considered to the most pathogenic due to involvement of the liver.2,3
Clinical signs of Eimeria infections are variable depending on the severity of infection. Rabbits may only suffer from growth retardation in mild infection whereas the disease may be fatal in severely infected cases, such as the case in this report. Heavily infected cases may show signs related to hepatic pathology, such as dullness, anorexia, debilitation, diarrhea, and/or constipation.1,2 Hepatomegaly, ascites and icterus are also commonly observed.4 Changes in the serum chemistry results may differ, depending on the disease stage. The initial stage may have evidence of hepatocellular injury (increased AST and ALT activities), followed by cholestasis (increased bilirubin).4,5 Increased GGT has been seen in experimentally infected rabbits4 and this may be associated with biliary hyperplasia (or cholestasis). At the end stage of disease, evidence of hepatic dysfunction (hypoglycemia) may be seen.5 The hypoglycemia seen in the present rabbit is likely associated with decreased gluconeogenesis due to hepatic failure from parasitism with a potential contributing component from anorexia. The diagnosis is usually readily made based on the identification of oocysts in bile aspirates or fecal examinations (Question 3), but histopathologic examination can be performed in animals that die suddenly.1,5 Typical gross findings of hepatic coccidiosis in rabbits are characterized by multifocal to coalescing, white to tan, firm, slightly raised, occasionally cystic nodular foci, which correlates with histological lesions of proliferative cholangiohepatitis, as is seen in this case.5 The characteristic proliferative biliary changes and organisms on histopathologic evaluation of necropsied tissue, both of which were seen in this case, are pathognomonic of this disease.5
The life cycle of Eimeria stiedae starts with ingestion of the sporulated oocysts from infected feces that then undergo excystation in the duodenum under the influence of pancreatic enzymes. Liberated sporozites penetrate the intestinal mucosa and migrate to biliary epithelial cells via lymphatics or blood vessels and undergo schizogony in biliary epithelial cells. Merozoites invade adjacent epithelial cells and undergo gametogeny, which develop into microgametes and macrogametes. After being fertilized by a microgametes, the macrogametes develop into oocysts. Oocysts rupture from epithelial cells and are passed into the bile and eventually the feces.1,2,4 Fecal shedding may persist for 7 or more weeks postexposure.5 The prepatent period is approximately 15-18 days. Oocysts are resistant in the environment and contaminated premises and fomites may be a source of infective sporulated oocysts for several months.5 The control of hepatic coccidiosis relies on good hygienic practices and the administration of anticoccidial drugs in feed or water in commercial rabbit farms.3
- Jenkins, JR. Gastrointestinal disease, section two, Rabbit. In Ferrets, Rabbits, and Rodents Clinical Medicine and Surgery. 2nd Quesenberry KE and Carpenter JW. St Louis, MI: Saunders. 2002. pp 161-171.
- Gardiner CH, Fayer R, Dubey JP. An Atlas of Protozoan Parasites in Animal Tissues. 2nd ed., Washington, DC: Armed Forces Institute of Pathology. 1998. pp 20-30.
- Redrobe, SP. Comparison of toltrazuril and sulphadimethoxine in the treatment of intestinal coccidiosis in pet rabbits. Vet Record. 2010: 287-290.
- Cam Y, Atasever A, Eraslan G, et al. Eimeria stiedae: Experimental infection in rabbits and the effect of treatment with toltrazuril and ivermectin. Exp Parasitol. 2008: 119:164-172.
- Percy DH, Barthold SW. Pathology of Laboratory Rodents and Rabbits. 3rd ed. Ames, IA: Blackwell Publishing. 2007. pp 288-290.
Authored by: M Asakawa (clinical pathology resident) and T Stokol. The authors thank Drs. May Tse and Teresa Southard, who shared the gross and histopathologic findings of this case.