Interpretation
Marked macrophagic (granulomatous) inflammation with intralesional bacilli consistent with Mycobacterium spp.
Explanation
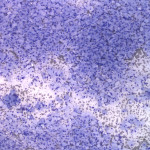
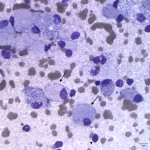
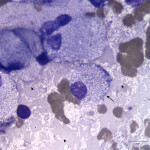
The examined smear was highly cellular consisting predominantly of macrophages (Question 1) in sheets with a few non-degenerate neutrophils and rare small lymphocytes on a proteinaceous background containing a moderate amount of blood. The predominance of macrophages is an abnormal finding, because aspirates from normal or hyperplastic lymph nodes should consist of mostly mixed lymphoid cells dominated by small lymphocytes (Question 1b), with only low numbers of other leukocytes present. The macrophages had markedly distended cytoplasm with abundant thin negative-staining bacterial rods (<0.5um wide x 3-5 um long), which were also noted extracellularly throughout the background of the smear. The morphologic appearance of the bacteria was consistent with Mycobacterium spp. (Question 2). This patient had risk factors for a mycobacterial infection including a nearly year-long history of immunosuppression for the previously diagnosed PRCA, as well as exposure to the outdoors and contact with wildlife (Question 3).
Additional information
Out of academic interest, a Ziehl-Neelsen (acid fast) stain was performed on an unstained cytologic smear from the jejunal lymph node. The organisms were strongly positive (staining red) confirming a mycobacterial infection. A sample from the lymph node was submitted to the National Veterinary Services Laboratories in Ames, Iowa for mycobacterial culture.
Discussion
Mycobacterial infections are caused by aerobic, non-spore forming, nonmotile, pleomorphic bacterial rods.1 Mycobacterium is a genus consisting of numerous species with a wide variety in host affinity and pathogenic potential.1 Mycobacteria of feline importance include (1) obligate pathogens (e.g. tuberculosis or TB), (2) mycobacteria that are difficult to grow on culture, so the environmental niche is unknown (e.g. feline leprosy syndrome (FLS), and (3) facultative pathogenic opportunistic saprophytes (e.g. non-tuberculous mycobacteriosis, NTM).2
Feline TB is recognized infrequently and is almost exclusively caused by M. microti or M. bovis.2 Feline infections with M. tuberculosis are very rare, because cats are naturally resistant to this species.2 Human beings are the only reservoir host for M. tuberculosis and spread disease via aerosolized droplets that are inhaled.1 Veterinary infections are considered an anthropozoonosis (transmission is from people to animal).1 Spread of M. tuberculosis back from dogs or cats to people has not been reported.1 Dogs and cats acquire M. bovis via ingestion of contaminated milk or meat.1 M. microti is predominantly a rodent pathogen that infects voles and has been seen most frequently in Great Britain.1 Cats can be infected with this species via ingestion of a small rodent (resulting in alimentary infection) or skin bites or wounds inflicted by an infected rodent (resulting in cutaneous disease). M. microti is less zoonotic than M. bovis; no cat to human spread of M. microti has been documented.2
Feline leprosy syndrome is caused by a number of mycobacterial species and is primarily a cutaneous disorder.3 Patients present with single or multiple, well-circumscribed, nodular granulomas in the skin and/or subcutis of the head, limbs, or trunk.2,3 Later in the course of disease, the infection may invade local lymph nodes or rarely spread systemically to internal organs (e.g. liver or spleen).2,3 Due to the inability to grow these species with routine culture techniques, molecular methods (e.g. PCR) are needed for confirmation.3
Non-tuberculosis mycobacteriosis is caused by many opportunistic mycobacterial species living in soil, water, and vegetation, and are ubiquitous worldwide.1,2 Fast or rapidly growing mycobacteria (RGM) (previously known as atypical mycobacteria) are most common, but slow growing variants are identified more frequently now with the use of molecular diagnostics.2,4 The RGM include the M. chelonae-abscessus, M. fortuitum, and M. smegmatis groups, as well as other groups and species.4 The RGM produce three syndromes in cats and dogs: most commonly (1) infectious panniculitis, (2) pyogranulomatous pneumonia, and rarely (3) disseminated systemic disease.4 Examples of the slow growing mycobacteria causing NTM are the closely related M. avium and M. intracellulare, which are referred to as the M. avium complex (MAC) due to the indistinct separation of these two species.1 Non-tuberculosis mycobacteriosis typically follows contamination of wounds or rarely entry through the gastrointestinal or respiratory tract.2
Mycobacterium avium complex appears to be more pathogenic than other NTM species and can cause a variety of clinical presentations including cutaneous lesions (mimicking TB or FLS), peripheral vestibular disease, generalized lymphadenopathy (including intestinal lymph node enlargement), pneumonia, gastrointestinal disease, intracranial infection, and often disseminated disease.2 Weight loss, anorexia, and fever have been observed.1 Infections with MAC in dogs and cats are often disseminated without indications of a primary granuloma at the site of entry.1 The organism is closely related to M. avium subspecies paratuberculosis, which is the cause of Johne’s disease in ruminants and other herbivores.1 Unlike M. tuberculosis and M. bovis, MAC organisms remain viable for at least 2 years in the environment, increasing the opportunities for disease transmission.1 The organism typically causes disease in birds, but can also infect human beings, dogs, and cats.1,2 Dogs and cats have innate resistance, therefore infections in these species is relatively uncommon.1 As an opportunist infection, MAC is usually seen in immunocompromised hosts and in cats may be associated with feline leukemia virus and feline immunodeficiency virus infection, toxoplasmosis, immunosuppressive drug treatment, or idiopathic T helper (CD4+) lymphopenia.1,2 Cats that hunt or fight are also at an increased risk of infection.2 No evidence has been found for spread of MAC among animals or people. Handling of dogs or cats infected with NTM is not considered a major health risk for immunocompetent people, because these bacteria are normally found in the environment in large numbers.1
Many cases of feline mycobacteriosis have a similar clinical presentation regardless of the causative species, therefore further testing for identification is needed, because the zoonotic risk, recommended treatments, and prognosis varies by species.2 The first step is confirmation of Mycobacterium spp., which often can be confidently performed on cytologic evaluation of tissue aspirates, as was done in this case. Mycobacterial organisms do not stain with Romanowsky stains due to the high lipid content of their cell wall giving them a characteristic cytologic appearance of nonstaining rods.1,5,6 The organisms are seen most often within macrophages, but are also often present extracellularly, as seen in this case.5,6 An acid-fast stain (e.g. Ziehl-Neelsen, Fite-Faraco) can be performed to aid in identification in suspect cases (for example with RGM infections where organisms may be much harder to visualize) or as a confirmatory stain when numerous organisms are present.4-6 The organisms appear as beaded linear bacteria with acid-fast staining.5 The high lipid content of mycolic acid in the mycobacterial cell wall is not only responsible for the absence of Romanowsky stain uptake, but also the acid-alcohol fastness.1 The absence of acid-fast staining does not rule out mycobacteria, as not all specimens will stain positive.1 Mycobacterial culture is the reference standard for diagnosis.1 Mycobacterial isolates can be identified by culture and specialized laboratories, such as the National Animal Disease Laboratory in Ames, Iowa, can determine their susceptibility to antimicrobial drugs.1 For mycobacteria that grow slowly or cannot be cultured, PCR on tissue specimens or body fluids can be performed to detect or identify organisms.2
Until the organism is identified, which can be as quick as 7 days with RGM or up to 12 weeks with the slow growing mycobacteria, it should be considered a potential zoonosis.1,2 Treatment is strongly discouraged for infected cats living with potentially immunosuppressed human beings. In cases where treatment is pursued, it is almost always long-term and may be indefinite. Treatment for confirmed M. tuberculosis or disseminated M. bovis is not recommended due to the potential human health hazard, and euthanasia is recommended.1,2 Infections with saprophytic mycobacteria, such as MAC and M. microti, would be the appropriate mycobacterial infections to consider treating in companion animal practice.1 Treatment depends on the distribution of disease and may involve surgical excision and/or multiple antibiotic therapy. Multidrug regimens are recommended to try and improve treatment success and avoid antimicrobial resistance, which can develop when single-agent therapy is used.1,2 The prognosis for mycobacteriosis depends on the species involved, as well as the extent and severity of the infection, and can widely vary.2
Case follow-up
In response to the cytologic diagnosis, the clinician discontinued cyclosporine and reduced the dose of prednisolone. Antibiotic therapy with clarithromycin, marbofloxacin, and rifampin was instituted. Antinausea medication (maropitant) was to be given as needed and an appetite stimulant (mirtazapine) was prescribed. Ultimately, the owners elected to euthanize the cat two weeks after the visit. Unfortunately, no organisms were isolated on the submitted mycobacterial culture. Given the clinical presentation of this case, including the history of immunosuppression and exposure to wildlife, along with the involvement of internal lymph nodes and absence of cutaneous lesions, infection with MAC or M. microti are considered to be the main likely causative organisms. In addition to the inability to culture FLS, M. microti has also resulted in failed cultures, as seen in this case.2
References
- Greene CE, Gunn-Moore DA. 2012. Infections caused by slow-growing mycobacteria. In: Greene CE, editor. Infectious diseases of the dog and cat. 4th ed. St. Louis, MO: Elsevier Saunders. pp 495-510.
- Gunn-Moore DA. Feline mycobacterial infections. The Veterinary Journal. 2014; 201: 230-238.
- O’Brien CR, Fyfe JA, Malik R. 2012. Feline leprosy syndromes. In: Greene CE, editor. Infectious diseases of the dog and cat. 4th ed. St. Louis, MO: Elsevier Saunders. pp 510-513.
- O’Brien CR, Fyfe JA, Malki R. 2012. Infections caused by rapidly growing mycobacteria. In: Greene CE, editor. Infectious diseases of the dog and cat. 4th ed. St. Louis, MO: Elsevier Saunders. pp 515-521.
- Raskin R. 2010. Skin and subcutaneous tissue. In: Raskin RE, Meyer DJ, editors. Canine and feline cytology: A color atlas and interpretation guide. 2nd ed. St. Louis, MO: Saunders Elsevier. pp 34, 37.
- Fisher DJ. 2014. Cutaneous and subcutaneous lesions. In: Valenciano AC, Cowell RL, editors. Cowell and Tyler’s diagnostic cytology and hematology of the dog and cat. 4th ed. St. Louis, MO: Elsevier Mosby. pg. 85.
Authored by: Drs. A. Newman (senior clinical pathology resident) and T. Stokol