Interpretation
Liver: Moderate mixed inflammation, necrosis, and cholestasis with protozoal organisms, presumptive Toxoplasma gondii.
Explanation
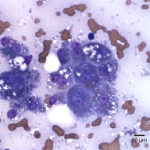
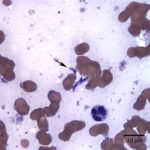
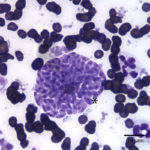
The liver aspirate was highly cellular and consisted of numerous sheets and clusters of hepatocytes admixed with a moderate number of mixed inflammatory cells. The hepatocytes often contained clear discrete vacuoles (compatible with lipid) and occasional binucleate forms were seen (Figure 1). The hepatocytes displayed moderate anisocytosis and anisokaryosis. There were numerous bile casts and intrahepatic aggregates of presumed bile pigment. Focal pockets of necrosis were also seen. The inflammatory cells consisted of mixed lymphocytes (these were mainly small lymphocytes with fewer intermediate and large forms), macrophages, and non-degenerate neutrophils. A few plasma cells were noted. Low numbers of approximately 1-4 μm zoites were seen extracellularly (Figures 1a, 2a) and within macrophages. The zoites ranged from crescent- to oval-shaped and had lightly blue cytoplasm and a central to paracentral round purple nucleus. The more oval forms were rarely seen in small extracellular groupings (Figure 3a). Morphologic features of the protozoal parasites were compatible with Toxoplasma spp.
Other protozoa (coccidia) with similar cytologic appearance to T. gondii in cats include Sarcocystis spp., Hammondia hammondi, and Besnoitia spp. Both Besnoitia spp. and H. hammondi, are thought to be non-pathogenic in cats and, aside from experimental studies, neither have been reported in extraintestinal tissues in cats.1–4 Only zoites of T. gondii and Sarcocystis spp. have been reported in extraintestinal tissue of naturally-infected cats. Therefore, T.gondii and Sarcocystis spp. were considered as differential diagnoses in this case (Question 2). However, because extraintestinal sarcocysts in cats have only been reported in cerebrospinal fluid (S. neurona) and in cardiac and skeletal muscle (Sarcocystis species not identified), T. gondii was considered the leading differential diagnosis.5–7,8,9
Discussion
Toxoplasma gondii is an obligate intracellular protozoan (coccidian) parasite that infects virtually all species of warm-blooded animals (birds and mammals). Felids, including domestic cats, can serve as both the definitive and intermediate host. The enteroepithelial cycle (coccidian phase) is found only in the definitive host, whereas the extraintestinal cycle (tissue phase) occurs in both felids and other intermediate hosts. Infections occur by three major pathways: 1) Ingestion of tissue containing cysts (bradyzoites and tachyzoites), 2) Ingestion of food or water contaminated with sporulated oocysts (with sporozoites being the infectious form) from definitive host feces, or 3) Congenital transmission (transplacental and lactational). In intermediate hosts, congenital transmission is transplacental only. Both transplacental and, less frequently, lactational transmission occurs in cats. Ingestion of tissue cysts containing bradyzoites is thought to be the main route of infection in cats.1,2 The explanation for the latter is that bradyzoites, but not tachyzoites, can survive the digestive process and more efficiently infect enteric epithelial cells.2 Additionally, bradyzoites are more infective to cats compared to oocysts.10 Once within enteric epithelial cells, bradyzoites differentiate into schizonts containing merozoites.11 Merozoites divide and form male microgamonts and female macrogamonts. Then, male microgametes budding from microgamonts fertilize the female macrogamonts. The resulting zygotes then mature into unsporulated oocysts, which are released into the environment, sporulate, and become infective.2,5 The extraintestinal cycle occurs after ingestion of sporulated oocysts or tissue cysts. Bradyzoites and sporozoites transit epithelial cells and enter nucleated cells within the lamina propria and replicate as tachyzoites within a parasitophorous vacuole.12 The tachyzoites are then disseminated via blood and the lymphatics and invade a wide range of cell types.13 Continued division of tachyzoites within infected cells, leads to cell rupturing, necrosis, and subsequent infection of new cells by released tachyzoites. Otherwise, with development of an antibody response, tachyzoites transform into slow-dividing bradyzoites and encyst. Tissue cysts form in the central nervous system, muscles, and visceral organs and probably persist for the life of the host. Immunosuppression allows tissue cysts to rupture and subsequent reactivation of infection.
Cats rarely show clinical signs during the enteroepithelial cycle of disease. Naïve cats, however, can develop-self-limiting diarrhea and young or immunosuppressed cats can simultaneously develop systemic disease. Clinical signs associated with systemic toxoplasmosis depend on which organs are affected, the degree of tissue necrosis, and the mode of infection. Post-natal infections are generally less clinically severe than congenital infections.1,2
The cat in this case demonstrated clinical signs commonly reported with post-natal toxoplasmosis, including lethargy, anorexia, and persistent hyperthermia.1,2 The mild icterus, abdominal discomfort, and ultrasonographic findings suggested liver disease. Liver disease was supported by biochemical results. Moderate to marked increases in ALT and AST activities were indicative of hepatocellular injury, whereas increased total bilirubin concentrations (with the direct form dominating over the indirect form) indicated cholestasis (Question 1). Liver aspirates confirmed hepatocellular injury (distended hepatocytes and pockets of necrosis (Figure 1) and cholestasis (bile casts). Cytologic examination of the liver aspirates also revealed the underlying cause in this case, with both individual zoites (Figures 1a and 2a) and cyst forms (Figure 3a) of the organism identified. Tachyzoite replication within infected hepatocytes likely resulted in hepatocellular necrosis and secondary inflammation. The presence of protozoa in the liver suggested systemic toxoplasmosis in this cat. Systemic toxoplasmosis is rarely reported in older cats but can result from spread of tachyzoites after initial acute exposure or reactivation of a chronic encysted infection due to immunosuppression.1,2,14 In this case, it is possible that immunosuppression was induced or exacerbated by the glucocorticoid therapy.
The CBC abnormalities, including the mild nonregenerative anemia, were attributed to inflammation. Both inflammation and nonregenerative anemia are non-specific findings reported with T. gondii infection.1,2
There is no serologic test that definitively confirms a diagnosis of toxoplasmosis. A tentative diagnosis can be made based on serum IgM and IgG antibody titers against T. gondii.1,2 A fourfold increase in IgG titer over a 2-3 week period or an IgM titer greater than or equal to 1:256 is indicative of active infection. Polymerase chain reaction (PCR) for the detection of protozoal DNA can be used to definitively confirm the presence of T. gondii in serum, tissue or cerebrospinal fluid (Question 3).15
Further information and follow up
Fine needle aspirates of the enlarged jejunal lymph nodes revealed reactive lymphoid tissue with an heterogeneous population of lymphocytes (these were primarily small lymphocytes with fewer intermediate and large forms) admixed with inflammatory cells (macrophages and neutrophils). Rare macrophages were seen phagocytizing similar protozoal organisms to those seen in the liver aspirate. These findings confirmed systemic T. gondii infection in this cat.
The cat was treated with antibiotics and intravenous fluids and the body temperature gradually normalized overnight (101.9°F). The next day, however, the cat’s condition progressively worsened and the cat developed neurologic signs and became hypothermic (95.7°F). Despite continued treatment and additional oxygen therapy, the cat went into respiratory arrest. Attempts at resuscitation were unsuccessful and the cat passed away. Further diagnostic tests, such as PCR or serologic testing, were not done.
References
- Dubey JP, Lindsay DS, Lappin MR. Toxoplasmosis and Other Intestinal Coccidial Infections in Cats and Dogs. Vet Clin North Am – Small Anim Pract. 2009;39(6):1009–34.
- Dubey, J. P., Lappin MR. Toxoplasmosis and Neosporosis. In: Green CE, editor. Infectious Diseases of the Dog and Cat. 4th ed. St Louis, MO: Elsevier; 2012. p. 806–27.
- Kirkpatrick CE, Dubey JP. Enteric coccidial infections. Isospora, Sarcocystis, Cryptosporidium, Besnoitia, and Hammondia. Vet Clin North Am Small Anim Pract. 1987;17(6):1405–20.
- Dubey JP, Sreekumar C, Lindsay DS, Hill D, Rosenthal BM, Venturini L, et al. Besnoitia oryctofelisi n. sp. (Protozoa: Apicomplexa) from domestic rabbits. Parasitology. 2003;126(Pt 6):521–39.
- Irvine KL, Walker JM, Friedrichs KR. Sarcocystid organisms found in bile from a dog with acute hepatitis: A case report and review of intestinal and hepatobiliary Sarcocystidae infections in dogs and cats. Vet Clin Pathol. 2016;45(1):57–65.
- Dubey JP, Sykes JE, Shelton GD, Sharp N, Verma SK, Calero-Bernal R, et al. Sarcocystis caninum and Sarcocystis svanai n. spp. (Apicomplexa: Sarcocystidae) associated with severe myositis and hepatitis in the domestic dog (Canis familiaris). J Eukaryot Microbiol. 2015;62(3):307–17.
- Dubey JP, Chapman JL, Rosenthal BM, Mense M, Schueler RL. Clinical Sarcocystis neurona, Sarcocystis canis, Toxoplasma gondii, and Neospora caninum infections in dogs. Vet Parasitol. 2006;137(1-2):36–49.
- Bisby TM, Holman PJ, Pitoc GA, Packer RA, Thompson CA, Raskin RE. Sarcocystis sp. encephalomyelitis in a cat. Vet Clin Pathol. 2010;39(1):105–12.
- C. E. Klrkpatrick, J. P. Dubey, M. H. Goldschmidt JES and JAS. Sarcocystis sp. in muscles of domestic cats. Vet Pathol … [Internet]. 1986;1–4. Available from: http://vet.sagepub.com/content/23/1/88.short
- Dubey JP. Comparative infectivity of oocysts and bradyzoites of Toxoplasma gondii for intermediate (mice) and definitive (cats) hosts. Vet Parasitol. 2006;140(1-2):69–75.
- Speer CA, Dubey JP. Ultrastructural differentiation of Toxoplasma gondii schizonts (types B to E) and gamonts in the intestines of cats fed bradyzoites. Int J Parasitol. 2005;35(2):193–206.
- Speer C a, Dubey JP. Ultrastructure of early stages of infections in mice fed Toxoplasma gondii oocysts. Parasitology. 1998;116 ( Pt 1:35–42.
- Dubey JP, Speer CA, Shen SK, Kwok OC, Blixt JA. Oocyst-induced murine toxoplasmosis: life cycle, pathogenicity, and stage conversion in mice fed Toxoplasma gondii oocysts. J Parasitol. 1997;83(5):870–82.
- Spycher A, Geigy C, Howard J, Posthaus H, Gendron K, Gottstein B, et al. Isolation and genotyping of Toxoplasma gondii causing fatal systemic toxoplasmosis in an immunocompetent 10-year-old cat. J Vet Diagnostic Investig [Internet]. 2011;23(1):104–8. Available from: <Go to ISI>://000286953200016\nhttp://www.embase.com/search/results?subaction=viewrecord&from=export&id=L361121060\nhttp://www.jvdi.org/cgi/reprint/23/1/104.pdf\nhttp://sfx.galib.uga.edu/sfx_emu1?sid=EMBASE&issn=10406387&id=doi:&atitle=Isolation+and+genot
- Burney DP, Lappin MR, Spilker M, McReynolds L. Detection of Toxoplasma gondii parasitemia in experimentally inoculated cats. J Parasitol [Internet]. 1999;85(5):947–51. Available from: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=10577732
Authored by: D Hernandez (clinical pathology resident) and T Stokol.