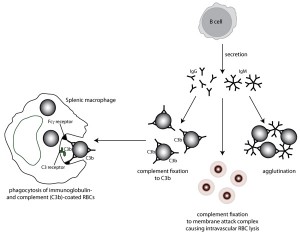
Auto-reactive B cells secrete immunoglobulins (IgG and/or IgM) that recognize a “self” epitope on red blood cells. Strong complement-fixing antibodies result in formation of the membrane attack complex (C6-C9), which punches holes in the red blood cell membrane causing them to rupture within the circulation, releasing hemoglobin and resulting in hemoglobinemia and hemoglobinuria (intravascular hemolysis). Weaker complement-fixing antibodies only generate the opsonin C3b, which also attaches to RBC membranes. Ig- or C3b-bound red blood cells are destroyed by macrophages (which contain receptors for C3b and the Fc portion of immunoglobulins) as they traverse through organs such as the spleen (extravascular hemolysis). Multivalent antibodies, such as IgM, can crosslink adjacent red blood cells, resulting in agglutination.