Interpretation
Mixed neutrophilic, histiocytic inflammation with intracellular carbon compatible with smoke inhalation
Explanation
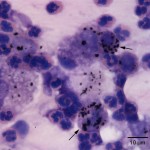
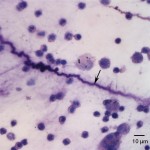
Direct smears were of high cellularity due to large numbers of non-degenerate neutrophils with fewer but still numerous macrophages also present. Macrophages were highly vacuolated, containing moderate to abundant amounts of black granular pigment (Figure 2a). The pigment was negative on a Prussian blue stain, and thus was presumed to be carbon (Prussian blue detects iron in hemosiderin) (Question 2). This was further supported by the absence of both hemosiderophages and erythrophages, which would indicate previous and ongoing hemorrhage, respectively. The background contained thick strands of mucous spirals (Question 3), also known as Curschmann’s spirals (Figure 3a). These could indicate increased mucus production with inspissation in airways and may be a consequence of the smoke-induced inflammation. No infectious agents were identified.
The findings were compatible with a mixed neutrophilic and histiocytic inflammatory response secondary to burn-related tissue injury and smoke inhalation (Question 1).
|
|
Case Follow-up
Subsequent transtracheal washes failed to reveal any bacteria present. The patient was again stabilized and remained so until the seventh day of hospitalization, when the mare rapidly deteriorated. The mare displayed severe respiratory effort, bilateral wheezes, and a marked decrease in respiratory rate. Endoscopic examination was performed and revealed abundant hemorrhagic and necrotic debris occluding the lower airways from the site of the tracheostomy. In spite of treatment, the patient failed to recuperate and continued to showed marked respiratory distress. Thus euthanasia was performed. Histopathologic examination of necropsy tissues revealed mucin with many macrophages in the bronchioles, as well as substantial anthracosis (correlating with the previously described cytologic findings).
Discussion
Smoke inhalation injury is associated with closed-spaced fires. The nature of the insult depends on the type of burned fuels, completeness of combustion, and generated heat intensity. There are three primary mechanisms of injury: direct thermal effects, toxic gas injury, and hypoxia. Direct thermal injury is the result of smoke contact with the face and mucosa of the upper airways, resulting in mucosal burns and upper airway edema and is most profound 18-24 hours after the fire. These effects are limited to the to the upper respiratory tract by laryngeal reflexes and efficient heat exchange within the nasopharynx and oropharynx, where superheated air is cooled before entering the lower respiratory tract.1-4
There are three categories of toxic inhalants: asphyxiants, systemic toxins, and irritants, which can induce damage directly and indirectly through inflammatory mediators.1,2 Carbon monoxide is an asphyxiant of major importance in smoke inhalation injury.3 It combines with hemoglobin forming carboxyhemoglobin (hemoglobin has 200-250 times greater affinity for carbon monoxide as compared to its affinity for oxygen) which is unable to transport oxygen. Additionally carboxyhemoglobin shifts the oxygen dissociation curve to the left by decreasing oxygen release at the tissue level and causing tissue hypoxia.1,5 Oxygen utilization at the cellular level is also affected by the competitive inhibition of carbon monoxide for cytochrome oxidase (impairing aerobic metabolism).5 Carbon monoxide also binds to myoglobin, thereby impairing oxygen transport to muscles.3 Other combustion products, such as hydrogen cyanide, hydrochloric acid, phosgene, sulfuric acid, and aldehydes, may induce severe tracheobronchitis, increased pulmonary pressure, peribronchial edema, mucosal sloughing, dysfunction of mucociliary apparatus, bronchoconstriction, and altered surfactant action. These changes can lead to the development of bronchopneumonia.4 With combustion there is also consumption of oxygen, leading to a decreased PaO2. The resulting low PaO2 can lead to pulmonary vasoconstriction and generalized hypoxia.1,2
In the horse, pulmonary dysfunction secondary to smoke inhalation includes three phases: acute pulmonary insufficiency, pulmonary edema, and bronchopneumonia.5 The first phase occurs while the horse is exposed to the deleterious environment created by the fire and during the first 36 hours following exposure. This phase is associated with lack of oxygen and excess carbon monoxide, as well as the depressant effect of smoke on the cardiovascular system.1,2,5 Irritating effects of noxious products, such as aldehydes, phosgene, and nitrogen oxides, as well as altered blood flow (10 fold increase in blood flow to the tracheobronchial tree) lead to the development of progressive edema and necrosis in the upper respiratory tract, followed by airway obstruction and bronchoconstriction in the lower respiratory tract.1,2 These insults facilitate the development of the second phase of smoke inhalation injury. This phase is characterized by pulmonary edema, lower airway obstruction, and pulmonary parenchymal lesions.1,2 Twenty four to 72 hours after exposure, pulmonary macrophages recruit neutrophils to the site of injury, where they release cytokines, proteolytic enzymes, and oxygen-derived free radicals. There is loss of microvascular integrity, leading to increased water in alveoli and impairment of gas exchange. Local injury also results in activation of coagulation and fibrin formation via the release of tissue factor. Cellular debris from the inflammatory cascade, fibrin, material deposited from smoke inhalation and neutrophils create pseudomembrane casts, which may attach to, and obstruct the small airways.1,2,5 Damage to the lower respiratory tract results in dysfunction of the mucociliary apparatus, and consequently reduced bacterial clearance, leading to the final phase of smoke inhalation injury. Bronchopneumonia occurs on average 7 to 15 days after the initial insult, and it most likely results from a combined effect of decreased bacterial clearance and increased susceptibility to infections due to local and systemic immunosuppression, such as from decreased function of alveolar macrophages.1,2,5
The onset of clinical signs may be delayed several days, thus a normal initial examination does not rule out exposure. Diagnostic tests should include a complete cell blood count, clotting profile, serum chemistry, urinalysis, arterial blood gas, carbon monoxide concentration, chest radiograph, endoscopy of the upper respiratory tract, and cytologic evaluation of tracheal aspirates.2,4 Due to the likelihood of edema and fluid shifts in these patients, packed cell volume, serum total protein and albumin should be frequently monitored.2
In transtracheal washes of normal horses, the predominant cells consist of ciliated epithelial cells, present individually or in small clusters, some of which are interspersed with low numbers of goblet cells.6–7 Alveolar macrophages dominate, with fewer numbers of neutrophils (<10% – this proportion may increase in stabled horses) and lymphocytes.6 Small numbers of binucleate or multinucleated macrophages can be present.7 Erythrocytes can be present, most likely as a consequence of minor trauma during sampling (this is less common in endoscopic samples). Aspirates tend to contain some mucus, appearing as thick stringy light blue amorphous strands, which sometimes form dark-staining tight spirals (Curschmann’s spirals). As indicated above, the latter (also called mucus spirals) are mostly seen in samples from animals with prolonged and excessive production of mucus.6 Contaminants from outside the lower respiratory tract, like squamous epithelial cells from the mouth or pharynx, fungal and plant material, may be found. Bacteria from the oral, pharyngeal, or nasal cavity or the environment may adhere to the surface of epithelial cells. Other foreign material rarely seen in washes include hairs, unidentified filaments, and pigmented debris.6,7 They may also represent inhaled material that has not been expelled by the mucociliary apparatus.6
After smoke inhalation, the epithelium of the trachea, bronchi, and bronchioli may be destroyed or severely damaged, resulting in inflammation, increased mucus production, edema, and bleeding into airways. On cytologic examination, inflammatory cells often consist of a mixed population of neutrophils and macrophages.8 Neutrophils are usually nondegenerate, however in nonseptic transtracheal wash samples features of neutrophil degeneration may be found. This can be attributed to the collection and handling of samples, particularly those left in the collection fluid for some time.6,8 Alveolar macrophages may contain erythrocytes and they are usually laden with black, carbon particles (anthracosis), which must be differentiated from hemosiderin via special staining techniques, as described above.6 Abundant mucus production may lead to the presence of Curschmann’s spirals, as seen in this case. Local and systemic immunosuppression and dysfunction of the mucociliary apparatus facilitate secondary bacterial and/or fungal infections, as well as decreased expelling of other inhaled substances, such as crystalline structures, pollen, and plant fibers.6
The mare in this case apparently progressed through at least two of the three described phases of pulmonary dysfunction secondary to smoke inhalation, including acute pulmonary insufficiency (at time of presentation), pulmonary edema (at approximately 24 hours after the initial insult). Euthanasia was performed seven days after admission, the described average time for bronchopneumonia to develop. It should be noted however, that chest radiographs were inconclusive for pneumonia and that both cytologic and histopathologic evaluation did not reveal a secondary bacterial infection. Most likely the mucociliary dysfunction, drying of lower airways and cough suppression as a consequence of the performed tracheostomy led to mucus and cellular debris accumulation within the lower airways. These factors and the progressive inflammation resulted in severe respiratory distress, prompting euthanasia.
References
- Marsh P. Fire and smoke inhalation injury in horses. 2007. Vet Clin N Am-Equine. 23. 19-30.
- Marsh P. Diseases of the Respiratory System: Smoke Inhalation, Chapter 31. In: Smith Bradford, ed. Large Animal Internal Medicine. 4th ed. St. Louis, MO: Saunders, Elsevier, 2009.
- McFarlane D. Smoke inhalation injury in the horse. 1995. J Equine Vet Sci. 15. 159-162.
- Hanson R. Management of burn injuries in the horse. 2005. Vet Clin N Am-Equine. 21. 105-123.
- Kirkland K. et al. Smoke inhalation injury in a pony. 1993. J Veter Emer Crit. 3. 83-89.
- Zinkl J. Lower Respiratory Tract, Chapter 5. In: Cowell R and Tyler R, ed. Diagnostic Cytology and Hematology of the Horse. 2nd ed. St. Louis, MO: Mosby, Inc, 2002.
- Whitwell K. et al. Collection and evaluation of tracheobronchial washes in the horse. 1984. Equine Vet J. 499-805.
- Burkhard M, Millward M. Respiratory Tract, Chapter 5. In: Raskin R and Meyer DJ, ed. Canine and Feline Cytology: a Color Atlas and Interpretation Guide. 2nd ed. St. Louis, MO: Saunders, Elsevier, 2010.
Authors: D Hernandez (clinical pathology post-doctoral associate), T Stokol
This case was part of a presentation by Kara Spillman as part of the Senior Seminar series.