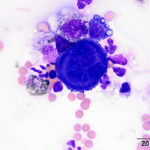
Pleural fluid from a horse
Case Information
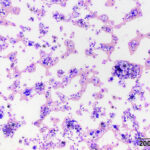
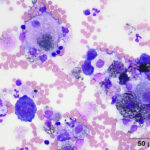
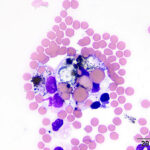
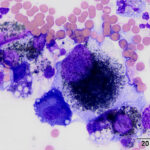
A 19 year old Arabian stallion was presented for evaluation for the cause of a pleural effusion. A large amount of pleural fluid was identified on thoracic ultrasonographic examination and the fluid was sampled for analysis. The fluid was dark yellow in color and slightly cloudy. Cell counts were low (800 nucleated cells/μL) with a small amount of blood (15.2 thousand RBCs/μL). Total protein by refractometry was mildly increased at 2.7 g/dL. Cytospin preparations of the fluid were prepared for cytological evaluation and were stained with a modified Wright’s stain.
Examine the provided images of the pleural fluid, then answer the following questions:
- How would you classify this effusion as to mechanism of formation?
- What other stains might you consider applying to this sample?
- What is your cytologic diagnosis?
Answers on the next page