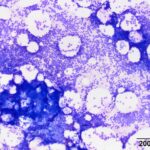
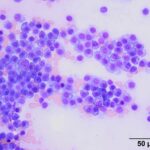
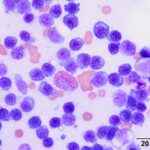
Bone marrow aspirate from a horse with hyperglobulinemia
Case Information
A 22-year-old Thoroughbred gelding presented for an approximately 3-week history of neurologic signs and neck stiffness. In addition, the horse was described to be pancytopenic and hyperproteinemic (8.3 g/dL; reference interval [RI]: 5.6 – 7.9 g/dL), due to a mild to moderate hyperglobulinemia (5.5 g/dL; RI: 2.4 – 4.7 g/dL). The albumin concentration was within the reference interval (2.9 g/dL; RI: 1.9 – 3.2 g/dL). The pancytopenia was characterized by a mild anemia (HCT: 22 %; RI: 30.0 – 47.0 %), leukopenia (3.7 K/μL; RI: 4.9 – 11.1 K/μL) due to a neutropenia (2.4 K/μL; RI: 2.5 – 6.9 K/μL) and moderate thrombocytopenia (49 K/μL; RI: 100 – 250 K/μL).
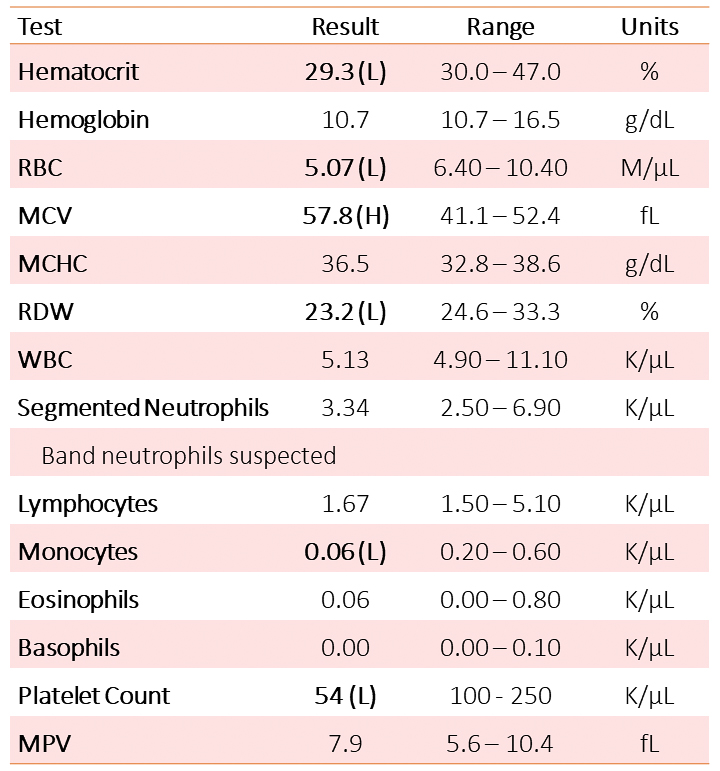
Upon re-examination by the veterinarian, a repeat complete blood count (CBC) was performed and results were as follows (Table 1):
 |
Given the CBC findings, a bone marrow aspirate and core biopsy was performed and submitted to the Animal Health Diagnostic Center (AHDC) at Cornell University. Direct smears of the freshly collected marrow were also prepared and submitted along with an EDTA-anticoagulated marrow sample.
Provided below re representative images of the bone marrow aspirate.
|
|
|
|
Using the provided information, answer the following questions:
- What is the main mechanism for the persistent anemia in this horse?
- Which is the predominant cell population in the provided images?
- What additional test would you recommend?
Answers on the next page.